 The Parabens, the Phthalates, the Silicones. These three cosmetic ingredients families are not chemically related and serve completely different purposes. Nevertheless, they all share the common point to be marked by the seal of infamy.
The Parabens, the Phthalates, the Silicones. These three cosmetic ingredients families are not chemically related and serve completely different purposes. Nevertheless, they all share the common point to be marked by the seal of infamy.
Abhorred by most of the journalists, products reviewers and, in general, by self-proclaimed experts, these ingredients have been blamed for every misfortune possible for years. From damaging the hair to causing breast cancers, from asphyxiating the skin to producing reproductive birth defects in babies. While it would be dishonest and irresponsible to claim that Parabens, Phthalates and Silicones are above reproach, it is obvious that these ingredients have been, for years, the object of a total and unfair misinformation. Some members of the families are truly infamous but where discernment was necessary, the market seems to have the perspective that all are unacceptable.
Is “free” the way to be?
Cosmetic brands started to label “free from” claims on their product labels and consumers got used to these new commercial arguments. With distance and hindsight, we all see today how bad was the idea to compete on the basis of safety. The day when all products are “Paraben free”, paraben will be dead and the competitive advantage of this claim will have vanished.
The bottom line is that the Cosmetic Industry is losing useful ingredients with no hope of return. Even worse maybe, the Cosmetic Industry also takes the risk to lose their alternatives. This happens when an ingredient is massively replaced by inappropriate substitutes, which in turn create real public safety issues.
A clear example is the replacement of safe parabens by Methylisothiazolinone (alone or in mixture with Methylchloroisothiazolinone). Both Methyl Paraben and Methylisothiazolinone are very effective preservatives but, unfortunately, the second one is a severe skin sensitizer. The explosion in demand for Methylisothiazolinone directly translated into a significant increase of the prevalence of skin sensitization to this allergen in the general population.
This increase did not go unnoticed by the European Authorities, who banned Methylisothiazolinone (alone or in mixture with Methylchloroisothiazolinone).1 For a number of reasons, the list of accepted preservatives is shrinking every year and there is no need for the cosmetic industry to accelerate this bad trend by shooting itself in the foot.
This article will summarize the regulatory status of Parabens; a follow-up article will address Phthalates and Silicones. We’ll also look at some viable alternatives. However, these alternatives should be considered with a critical eye. First because one-to-one substitutes are rare and the choice of an alternative is often a matter of compromise. Then because, as mentioned above, phasing out an ingredient is a serious decision that may finally make its use impossible. Look before you leap!
Parabens
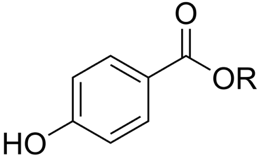 Parabens are a family of esters based on parahydroxybenzoic acid. All members of the families show strong antimicrobial properties and they are used in cosmetic products as preservative ingredients.
Parabens are a family of esters based on parahydroxybenzoic acid. All members of the families show strong antimicrobial properties and they are used in cosmetic products as preservative ingredients.
The ‘Paraben Crisis’ started in 2004 when a scientific article made a correlation between the presence of parabens in the human body and breast tumors.2 This controversial article got viral and was highlighted by the media3. From this point, Parabens became public enemy number one for many consumers and, very quickly, for many cosmetic brands too. Then the marketing claim “Paraben free” started to flourish everywhere. It should be noticed that this claim is considered as denigrating and should be prohibited soon at the European level.4
Parabens have been thoroughly evaluated5,6,7 by the Scientific Committee on Consumer Safety (SCCS), one of the most stringent scientific committees of the European Commission, and regulatory decisions were taken:
| Name | CAS | Status |
| Methyl Paraben | 99-76-3 | Restricted (V, 12)
· Max 0.4% for single ester (as acid) · Max 0.8% for the sum of esters (as acid) |
| Ethyl Paraben | 120-47-8 | Restricted (V, 12)
· Max 0.4% for single ester (as acid) · Max 0.8% for the sum of esters (as acid) |
| Propyl Paraben | 94-13-3 | Restricted (V, 12a)8
· Max 0.14% for the sum of Propyl Paraben and Butyl Paraben (as acid) |
| Isopropyl Paraben | 4191-73-5 | Banned (II, 1374) |
| Butyl Paraben | 94-26-8 | Restricted (V, 12a)8
Max 0.14% for the sum of Propyl Paraben and Butyl Paraben (as acid) |
| Isobutyl Paraben | 4247-02-3 | Banned (II, 1375) |
| Pentyl Paraben | 6521-29-5 | Banned (II, 1378) |
| Phenyl Paraben | 17696-62-7 | Banned (II, 1376) |
| Benzyl Paraben | 94-18-8 | Banned (II, 1377) |
In other words, the use of Methyl Paraben and Ethyl Paraben (and, in a lesser extent, of Propyl Paraben and Butyl Paraben) at effective concentrations is still legal and deemed safe. Despite all the disinformation that can be found on the internet, no direct links have been established between these parabens and cancer as of today.
Parabens have many advantages: they are effective against Gram+ and Gram- bacteria but also against yeast and mold at relatively low concentrations. Furthermore they are inexpensive and compatible with many product types although their optimum pH is typically below 7. They also have some drawbacks and, in particular, a recognized incompatibility with most of cationic and non-ionic surfactants, cellulose esters, PEG and gelatins.
Paraben alternatives
One of the best alternative is probably Phenoxyethanol (CAS 122-99-6), which can be used in the EU at up to 1.0 % in all product categories. Its safety has been re-confirmed recently by the SCCS9 and no regulatory uncertainty is identified at mid to long term.
The optimum pH of Phenoxyethanol is between 4 and 9 and, like the parabens, it has the advantage to be inexpensive and the disadvantage to be incompatible with most of the non-ionic surfactants. However, despite its qualities, Phenoxyethanol is mainly active against Gram- bacteria. Like the Parabens, it is active against a broad spectrum of microorganisms but its activity against Gram+ bacteria, yeast and mold is not always totally convincing.
Sodium Benzoate (and Benzoic Acid), potentially in combination with Potassium Sorbate, is also an acceptable alternative. Sodium Benzoate can be used at the following levels:
- up to 2.5% in rinse-off products
- 1.7% in oral care products
- 0.5% in leave-on products
- 0.06% and above shows activity against yeast and mold
Sodium Benzoate (and Potassium Sorbate) are inexpensive and can be easily sourced from nature (and therefore have the advantage to be compatible with a natural certification). However, the ideal pH for Sodium Benzoate alone or in combination with Potassium Sorbate is mainly between 3 to 5, which could be a problem, because many products are formulated out of this range. Using this preservative would be a mistake, as its efficacy could be low or even negligible. Furthermore, Sodium Benzoate is not a broad-spectrum preservative and usually has to be combined with a preservative active against Pseudomonas (Gram- bacteria).
Similarly, Benzyl Alcohol can be a valid alternative. Active against Gram+ bacteria from 25 ppm, it will generally be combined with Dehydroacetic Acid, more active against yeast and mold. Benzyl Alcohol can be used at up to 1.0% and Dehydroacetic Acid at up to 0.6%. This combination will be preferably used at a pH between 3 and 5. Incompatibility is low except with the non-ionic surfactants and this combination can also be sourced from nature.
Preservative-free cosmetics?
 Other convincing alternatives exist and this article does not seek to list them all. However, it is worthwhile to highlight the possibility to formulate preservative-free cosmetic products. The most common option in skincare products is the use of airless devices, such as the airless pump bottles by The Packaging Company, combined with a rigorous quality control system to avoid microbiological contaminations during the manufacturing process.
Other convincing alternatives exist and this article does not seek to list them all. However, it is worthwhile to highlight the possibility to formulate preservative-free cosmetic products. The most common option in skincare products is the use of airless devices, such as the airless pump bottles by The Packaging Company, combined with a rigorous quality control system to avoid microbiological contaminations during the manufacturing process.
The cosmetic product is then formulated without preservatives, which is acceptable as the finished product and the packaging are completely sterile and not in contact with the external environment.
The packaging is clearly more expensive than traditional pumps or jars but it gives access to interesting competitive advantages such as the marketing claim “Preservative free”. Please note that “Preservative free” is 100% EU compliant while “Paraben free” is not.
Further reading:
- Consumer Concern Over Parabens Leads to Trending Alternatives
- The Bad, the Banned, and the Maligned: Formulating with Your Hands Tied
- The Challenge of Natural Cosmetics
- Preservation Strategies for Global Personal Care Formulations
References:
- Methylisothiazolinone is prohibited by the EU Cosmetics Regulation (EC No 1223/2009) in leave-on cosmetic products and will be restricted at only 15 ppm in rinse-off cosmetic products in the next few months. Kathon CG is also prohibited in leave-on products and is already restricted at 15 ppm in rinse-off products.
- Concentrations of Parabens in Human Breast Tumors by P.D. Darbre, A. Alijarrah, W.R. Miller, N.G. Coldham, M.J. Sauer and G.S. Pope, Journal of Applied Toxicology, 24, 5-13 (2004).
- g. “Envoyé Spécial”, airdate March, 3rd, 2005
- Report from the Commission to the European Parliament and the Council on product claims made based on common criteria in the field of cosmetics [PDF] (see section 3.3.3)
- Scientific Committee on Consumer Safety (SCCS) Opinion on Parabens COLIPA n° P82 [PDF]
- SCCS Opinion on Parabens UPDATED request for a scientific opinion on propyl- and butylparaben COLIPA n° P82 [PDF]
- Scientific Committee on Consumer Products (SCCP) Extended Opinion on the Safety Evaluation of Parabens [PDF]
- Prohibited in leave-on products designed for application on the nappy area of children under three years of age.
- SCCS OPINION ON Phenoxyethanol [PDF]
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



We are very happy to read this, thank you for the great insights, we have been fighting this fight for a long time and are terribly affected by irresponsible media releases like the one published in 2004. Consumers need this information to make informed decision. Se use Parabens, but in very very low percentages and are discriminated against in the EU market.
Thank you for the very interesting article.
I wish I had known about the ban on Methylisothiazolinone in Europe. This preservative is still being used in the USA.
Recently my two dogs suffered horrible skin lesions after using a US made dog shampoo – the most-sold brand in supermarkets. Why has the FDA not banned it yet?
Questions like this are exactly why formulators have so many problems. Spurious logic, decisions made with no basis in fact whatsoever. “I read online that MIT can cause skin sensitivity issues. I have a pet who used a product containing MIT, and had a skin issue. Why isn’t MIT banned?!?!”
The internet is a fantastic thing. It is incredible to have such a wealth and volume of information available at any time. But it now allows people to believe they are experts in all fields! In the end, it just makes things worse for everyone.
Cosmetic industry regulation in the EU is way more strict and regulated than in the U.S.
As formulator ,(but also as an avid consumer), I always refer to European regulations.
The U.S. companies sell products globally therefore use the strictest standards like that of the EU when formulating products. Also to add to the MIT/MCI facts…MIT was banned in Leave-on products. It has been tested extensively by SCCS which is EU’s research organization of scientist and found that at CERTAIN levels there is no skin sensitivity in rinse off products. That is why its not banned. These safe limits are what companies are/may use. If it is in your rinse off products especially by major companies it is being used within safe limits. When reading literature that indicates to the contrary keep in mind a few things. Those articles don’t mention they are not safe for use or cause skin irritation ABOVE a certain level. They often take the information and don’t complete the sentence. MIT is not safe for use….ABOVE 15ppm in rinse off products. If you read an article that leaves off the second part of that statement question the writer of that article. The EU is looking at human skin not canine skin so there maybe something to that doggie concerns. If your dog reacts use something else. You and Your children are safe with the limits that have been set which are 15ppm.
The distinction is critical because, as was noted, the tool bag of preservatives formulators and microbiologist have to protect us from harmful bacteria is very limited. Job #1 is to protect and Preservation=Protection!! “Preservative” is not a bad word. FDA has made it clear, products claiming to be preservative free are a RED FLAG for them. Let’s stay informed and not fall victim to the headlines that are ill informed. The cosmetic industry and microbiologist within it share information, are mothers, and fathers, and just like you want safe products. They work together as scientist, ensuring the industry is one of the best self regulated industries around. what they work with is accurate scientific facts and data that they test and can be repeated globally. They share that information outside of their companies purposely so consumers of their products are safe! This is not an us vs. them situation. They are us! They too use the products and have families to keep safe. They work tirelessly to keep you safe, lets not take the tools away that help them do that because someone without the complete facts claims to know better. Lets just be informed, and know often those articles don’t give you complete information. In the case of MIT it is safe for use up to 15ppm in rinse off products. An article or blog claiming that its not safe for use isn’t completely accurate. It isn’t safe for us ABOVE 15ppm in rinse off.
Thanks for your article on preservatives very educative & informative, please i need to the broad spectrum of preservation on combination of methylisothiasolinone & benzisothiasolinone.
Thanks .
Godswill Nwokolo,
Lagos, Nigeria
Thanks for the article.
Though there are some points I can’t agree with:
1. “PARABENS have some drawbacks and, in particular, a recognized incompatibility with most of cationic and non-ionic surfactants, cellulose esters, PEG and gelatins.”
It’s better to add links to some peer-reviewed articles investigating that problem. Some articles that I know are:
S. M.Blaug “Interaction of Parabens with Nonionic Macromolecules” (1961) and R.D.Charles “THE EFFECT OF SORBIC ACID AND OTHER PRESERVATIVES ON ORGANISM GROWTH IN TYPICAL NONIONIC EMULSIFIED COMMERCIAL COSMETICS” (1959).
Blaug shows that parabens can interact with non-ionics in aqueous systems. From another side Charles stated that commercial cosmetics with parabens and non-ionics can successfully pass challenge tests. Hence Blaug made the right conclusion, that cosmetic systems are much more complex than water solutions, and preservation of cosmetics depends upon more factors than interaction between preservative and macromolecule.
2. Phenoxyethanol – “disadvantage to be incompatible with most of the non-ionic surfactants”. I spent several minutes trying to find any articles about that, but failed.
Phenoxyethanol and parabens can be used in cosmetics based on non-ionic surfactants. Though it doesn’t mean that you should not do some challenge tests.
So the term “incompatible” is a little bit misleading here, making the life of formulation chemist more complicated. Please do not discredit preservatives more, than they already are 🙂
Yes, it is still used in the USA. As a matter of fact, I have a very high priced shampoo with these preservatives in it. Thanks to the writer and publisher of this article for giving me insight as to what to look out for when purchasing my cosmetic products. Capitalist societies! Will settle for more of EU cosmetic products. Thank you.
Paraben free products, Replace the MP and PP what kind of preservatives use in Products please suggest
I am in fact allergic to MI. Allergy patch test proved it. So are numerous other people. So no it’s not safe for everyone. Skin is skin and yes that poor dog I know the feeling. I’m still suffering from the effects of using products with MI 3 years later so yea this is why people give a crap with what they’re putting on their bodies thank you.
A good amount of people are (quite literally) deadly allergic to peanuts and tree nuts. Why are they not banned? Because even though it’s not safe for THEM, it is perfectly safe for the the VAST MAJORITY of people. And the effects are in fact, temporary and discontinuing exposure should be enough. When it comes to cosmetics, the same applies. If the rash/itchiness persists you need to see a doctor, as you may have an underlying condition.
Allergies are a very personal condition as you can see. When formulating products, scientists need to make trade offs. Do I create a product that causes no adverse reactions on 90% of the population but is safe for everyone? Or do I risk under-preserving my product and potentially allowing growth of deadly bacteria so I can cater to everyone?
So you get an idea of how important preservatives are. Even if a product is made in sterile conditions, once a container leaves the manufacturer you have zero control over what happens next. People do unsafe things, products get contaminated during normal day to day use, and without the right preservative you may be allowing for growth of bacteria that may cause much more damage than some itchiness on 10% of the population.
That been said, the fact that a product causes a skin reaction does not mean it’s not safe. Lemon juice, pineapple, potatoes, and an infinite number of things cause skin reactions, worse on some people than others. I don’t see them being banned anytime soon.
Unsafe things people do: cut open a tube of cream or other cosmetic when it’s about to run out, dilute shampoo or body wash, pick up a product from a jar with something non-sterile (fingers, spoon, brush). Bacteria live everywhere, and some of them are quite dangerous but our bodies can fight them when in small amounts. Some cosmetics, however, are the perfect breeding ground for bacteria and the issues from unpreserved or underpreserved cosmetics can cost lives.
In my opinion, I would rather risk a small number of people getting rashes that WILL go away than a single dead person.
I’m delighted to tell that your article is extremely interesting to read. I never stop myself from expressing my dissatisfaction with it. You’re doing an outstanding job. Keep going.
What an amazing post!! This is jam-packed with excellent material, and I can’t wait to delve in and begin utilising the tools you’ve provided. Your euphoria is invigorating.