 Formulating hydrophobic (HP) and superhydrophobic (SH) coatings has a myriad of challenges and considerations. There are multiple means to achieve a temporary SH surface that provides a high contact angle as well as a low roll off angle, however, the challenge is to formulate a long lasting material that maintains its performance for an extended period of time that includes abrasion resistance, light stability, and resistance to a variety of environmental conditions for the intended application. A well-designed HP or SH coating for the intended application can provide a variety of benefits for applications in addition to water repellency that can include:
Formulating hydrophobic (HP) and superhydrophobic (SH) coatings has a myriad of challenges and considerations. There are multiple means to achieve a temporary SH surface that provides a high contact angle as well as a low roll off angle, however, the challenge is to formulate a long lasting material that maintains its performance for an extended period of time that includes abrasion resistance, light stability, and resistance to a variety of environmental conditions for the intended application. A well-designed HP or SH coating for the intended application can provide a variety of benefits for applications in addition to water repellency that can include:
- Corrosion resistance
- Self-cleanability
- HP/SH sustainability after surface erosion
- Volume super-hydrophobicity rather than just surface hydrophobicity
- Stain resistance
- Resistance to microbe growth associated with dirt collection
- Anti-fouling marine coatings
- Low water-drag marine coatings
- Protective coatings for harsh environments such as oil rigs
Superhydrophobic surfaces in nature
There are reported to be thousands of examples of SH bio-formed materials found in nature. Some of the common ones include lotus leaves, rice leaves, duck feathers, rose petals, and the legs of water-striders. The surface of these naturally occurring biobased materials all have a patterned microstructure combined with a layer of a low surface energy substance. The lotus leaf has a microstructure comprising small protuberances or spiked papillae 10 – 20 microns in height and 10 – 15 microns and the distances between the nearest papillae is about 15 microns. The tips of the papillae are coated with a hydrophobic wax layer.

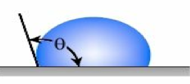
Accordingly, due to the super-hydrophobicity of the lotus leaf surface, the water cannot penetrate the valleys between the wax-coated papillae so the water droplet forms a contact angle of 150 degrees or higher (figure 4). A hydrophobic surface is defined as a surface with a static contact angle of > 90 degrees (figure 3). Hydrophobic (repels water) or Superhydrophobic coatings (highly water-repellent) can provide a variety of beneficial properties as stated above. Several factors impact the contact angle of a water drop on the surface of a coating. These include the macro, micro, and nano-surface profile, and the surface tension of the coating on which the water droplet is resting. Surface tension is the elastic tendency of liquids that make them acquire the least surface area possible. The equation that defines this in relation to a solid surface is known as Young’s equation:

Young’s equation demonstrates that hydrophobicity can only be observed on solid surfaces that provide a low σsv.
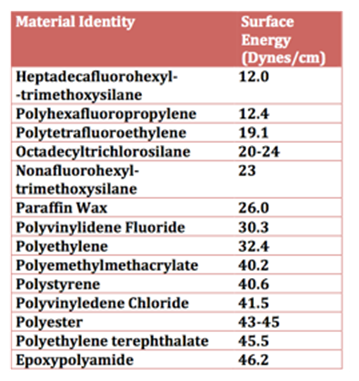
To maximize the surface hydrophobicity of a coating, the surface energy should be as low as possible. Low surface energy, coupled with an appropriately structured surface, maximizes hydrophobicity. Surface energy has the same units as surface tension (force per unit length or dynes/cm). A high surface tension liquid such as water will have maximum hydrophobicity and thus have poor wetting (high contact angle) over a coating surface that has low surface energy. As Table I illustrates, surface energy can vary greatly depending on the nature of the surface that comes in contact with water.
Table I – Surface Energy of Materials
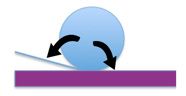

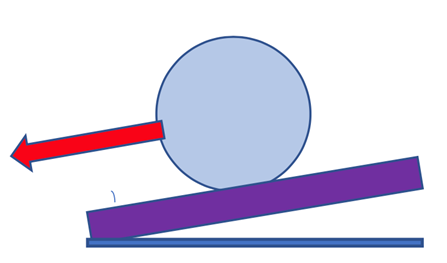
In Figure 6, for the Lotus leaf, the water droplet only touches the tips of the papillae and traps air between the Papillae resulting in a low adhesive force resulting in a high contact angle, and a low roll off angle.
Formulating Hydrophobic/Superhydrophobic Coatings
The evolution and development of HP and SH coatings were inspired by nature but utilize synthetic materials. Accordingly, SH coatings developed in the lab mimic many of the same attributes of SH surfaces in nature such as combining an appropriate micro-surface structure along with the use of low surface energy materials on or at the tips of spiky surfaces (Fig. 2). Some of the structured materials used in coatings include Hydrophobically or SH-modified pigments such as fumed silica, diatomaceous earth and finely ground silica can produce bulk hydrophobicity rather than just surface hydrophobicity. Surface hydrophobicity is normally a few nanometers thick. The advantage of a coating with bulk or volume hydrophobicity provides long-lasting hydrophobicity even when the surface of the coating is eroded (Fig.7). An HP or SH coating surface can lose HP or SH surface properties relatively quickly when put into service, whereas a coating that has volume or bulk HP/SH properties as well can maintain these properties over a longer time frame. Some examples of materials that can provide hydrophobic surface properties to coatings include waxes, oligomers, or polymers added in low percentages which may stratify at the surface but are more prone to erode away when the coating is placed into service. Other means to improve hydrophobicity include low surface tension reactive oligomers, polymers containing a high percentage of HP monomers, and modifying paints, resins, and pigments with silanes and siloxanes which provide a low surface tension.
A search of Raw Materials on the Prospector web site using the word hydrophobic followed by the material category (e.g. pigments, monomers, oligomers etc.) will provide a variety of Hydrophobic materials

Surface modification of pigments to provide a very low surface energy can have a dramatic effect on hydrophobicity and the proper reactant or surface modification can provide the modification to enable SH performance. Many pigment surfaces can be surface modified with a silane functional group that reacts with or is absorbed on the pigment surface.
For example, a coating comprised of polyhexafluoropropylene (12.0 Dynes/cm) on the surface will provide a more hydrophobic surface than that of polymethylmethacrylate (40.2 Dynes/cm). In general, terms, to provide the greatest surface hydrophobicity, the material’s most hydrophobic moiety should be positioned on the surface. Perfluoro and aliphatic groups at the coating surface offer greater hydrophobicity than that of ester or alcohol groups. For example, from lowest to highest surface tension:

Providing increased hydrophobicity throughout a properly engineered coating can also provide additional attributes such as improved corrosion and moisture resistance.
In summary, several key principles for considering the design and formulation of hydrophobic and superhydrophobic materials and coatings were briefly described in this article. A few challenges remain, for example, one of the important considerations is to obtain and maintain the proper balance of SH properties with that of the mechanical surface strength of the coating and an acceptable balance of other physical, chemical, and accelerated properties.
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.


