Jon Dekar is only 31 years old, but he has spent a decade researching and developing a product he calls Obi, and the past three years further refining it and scaling up production. Obi is a tabletop robotic device designed to help restore a degree of freedom and dignity to individuals who cannot feed themselves.
Dekar –– a mechanical engineering graduate from the University of Dayton in Ohio –– prefers to call Obi a “robotic dining companion.” Indeed, he said in a recent interview, “kids tend to personify it the most, and treat it like a pet.”
Last year, Obi won a 2018 Gold Medical Design Excellence Award in the category for Rehabilitation and Assistive-Technology Products. But for Dekar, the real prize is seeing the very human and emotional reactions from users who, in many cases, are able to feed themselves for the first time. It’s not uncommon, he notes, for all involved to see tears of joy.
Recognizing a market need

Dekar began seriously researching this topic of assisted feeding during his first year in college, after seeing a degenerative disease rob his paternal grandfather of the ability to eat under his own power. Dekar was astounded to find there were no truly effective devices on the market to address this dilemma. He applied his engineering knowledge, and tapped into the business acumen of his father, Tom, who is also an engineer but who worked as a management consultant. Together, they mapped out a business plan.
Dekar didn’t want to create just a feeding robot, he wanted to design something cool, something that looked friendly and that had personality. He launched his company, DESĪN LLC, and got to work. In 2016, after years of research, trial and error, testing and prototyping, Obi was born. Obi’s name comes from the word “obeisance,” which refers to a gesture of respect or deference.
“I was inspired by the Disney Pixar lamp,” Dekar told this reporter at the time of the product’s initial launch in July 2016. The aim, he said, was to inject some whimsy. “For people to really engage with robotics, I believe that [the device] has to have a subtle personality, to make it more approachable…so that instead of being fearful of the robot, you become charmed by the child-like personality of the robot.” Indeed, Obi makes a little bow to the user when he’s powered on.
DESĪN (pronounced “Design”) has just launched the second-generation Obi, which is considered an FDA Class 1 medical device. Dekar has secured 10 U.S. patents, gained both UL certification and CE compliance, and now is distributing Obi in more than 20 countries.
Several different advances

For Gen2 of the all-white device, “Everything has been dramatically iterated forward,” he said in a Sept. 11 phone interview. Obi is still using most of the same polymer materials for the device’s 30 or so plastic parts, but DESĪN has modified the injection tooling to enable the production of snap-fit components, significantly upgraded the motors, added more robust environmental protections to all the electronics, and improved the cabling and connectors.
It is using two different grades of polypropylene –– a food-grade, custom PP compound from RTP Co. for the plate and four food bowls, and a slightly softer, off-the-shelf PP resin from Total Plastics for the utensils. DESĪN uses a Cycoloy-brand, medical-grade, flame-retardant PC/ABS from Sabic for non-food-contact enclosure parts, and a grade of Sabic’s Lexan LUX polycarbonate resin for the surfaces and rings around its LED lights and switches.
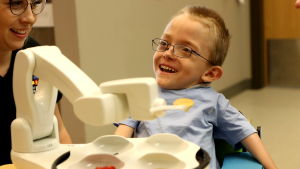
Obi also uses a soft-durometer Versollan thermoplastic elastomer from PolyOne Corp. on the overmolded bottom piece, which leverages the material’s high surface energy to help the device grip the table, to keep it from sliding. And Uniform Color Co. supplied the additives for the appliance’s all-white plastic parts.
Dekar, meanwhile, says he has devoted 70 percent of his time in the past year to setting up a 3,000-square-foot office and factory in Dayton, to allow DESĪN to do all of its engineering, testing, assembly, packaging and certification assessments in-house. The firm has partnered from the beginning with custom injection molder and mold maker PTI Engineered Plastics in Macomb, Mich., for all of Obi’s molded parts and tooling.

Tapping the institutional market
Dekar says he is expanding the market for Obi with a push into the institutional sector, such as nursing homes, hospitals, rehabilitation facilities, and the like. The product gets much rougher treatment in such shared environments, hence the need to make the device more robust overall.
As a result, he says, we’ve upgraded the motors to all-aluminum bodies (vs. the previous aluminum/ABS combination) to make them much more rigid. They now have four times the mechanical strength and twice the torque output as the previous version. They also run cooler, for a much longer period of time, and have a longer battery life.
Additionally, the previously noted improvements to what Dekar calls “environmental protections” have made Obi’s electronics more resistant to abuse or misuse. That includes such things as protecting against spills, and other factors such as temperature and humidity fluctuations through storage or use.
Finding a way to modify the tooling to enable production of snap-fit plastic components also was vital, as it saved huge sums of money from having to have PTI make all new molds. Dekar says he worked with PTI to develop a successful “part mating strategy” that made this possible.
Walled Lake, Mich.-based design firm Sundberg-Ferar, which helped Dekar to develop and refine the first-generation Obi, also has informally lent some mechanical support for redesigning the plastic parts to accommodate the tooling changes.
Smoother operation
From the user’s perspective, Dekar notes, the biggest improvement between the two versions is that the feeding arm’s movement is visibly smoother and offers twice the degree of precision.
Dekar says he is satisfied with Obi’s progress so far. “We’ve definitely proven there is a market for this. And we had to push it into the market, because there was no pull into the market.”
He declined to discuss specific sales numbers but says DESĪN is “now talking about selling thousands [of units] per year, not hundreds.”
Obi is priced at about $6,000 per device, but he notes that “nearly every product we sell is reimbursed” by some type of grant or state Medicaid program.

Still, the institutional market offers another benefit, besides large potential sales numbers. Those customers do not require reimbursement. In fact, institutional users can claim the device is actually a productivity aid for their business since, for example, it frees up skilled nursing personnel to handle other tasks rather than hand-feeding patients. Additionally, those organizations that adopt Obi potentially also can highlight it as a means to promote their business as being technologically advanced, and can even charge an additional fee for use of the device, if they wish.
Jon Dekar realized more than a decade ago that there was not a functional problem in moving food from point A to point B. Engineering could handle that. Rather, what he faced was a design problem. He set out to create a safe, functional self-feeding appliance that was also simple, intuitive, and cool enough that people would want to use it. Now, with Obi graduating to its second generation, signs are that he has succeeded in doing just that.
For more information on the device, go to www.meetobi.com.
Search @obirobot on Facebook for Jon’s work.
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



Leave a Reply or Comment