Introduction
 Most paints and inks contain solid particles, being pigments and/or fillers. Two instability problems, flocculation (the gluing together of separated particles) and sedimentation, can occur with solid particles that have been separated from each other and that are distributed in a paint or ink. In this article, learn about the basics of sedimentation and how to prevent it.
Most paints and inks contain solid particles, being pigments and/or fillers. Two instability problems, flocculation (the gluing together of separated particles) and sedimentation, can occur with solid particles that have been separated from each other and that are distributed in a paint or ink. In this article, learn about the basics of sedimentation and how to prevent it.
Sedimentation defined
Sedimentation is the process of solid particles sinking during storage because of gravity. In extreme cases, particles can cluster together on the bottom of the can because of sedimentation. This highly undesired phenomenon, resulting in the formation of a hard or rubbery sediment, is called settling.
The basics of sedimentation
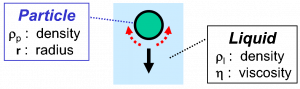
In general, solid particles in a liquid are pulled down because of gravitational force. The cause of this phenomenon is that many solids have a density that is higher than the surrounding liquid’s density. The process of particles sinking (sedimentation) causes problems during storage. The speed of sedimentation is governed by a few properties, shown in Figure 1.
The sedimentation velocity of a solid particle having perfect spherical shape in a Newtonian liquid, shown in Equation 1, was derived by Sir George Stokes.

In this equation ‘g‘ is the gravitational acceleration.
The most important factor, governing how fast a particle will sink, is the density of the particle, or, to be more precise, the difference between the density of the particle and the density of the surrounding liquid (ρp – ρl).
The second factor is size: big particles sink faster than small particles. The sedimentation velocity of a spherical particle is proportional to the square of the radius of the particle (r2).
The third factor is the viscosity of the surrounding liquid (η). A liquid with low viscosity results in faster sedimentation.
The equation is valid for smooth particles of spherical shape. A fourth factor, governing how fast a particle sinks, is the shape of the particle.
Preventing sedimentation
In order to understand how sedimentation can be prevented, take into consideration that sedimentation is mainly a problem that occurs during storage of a paint or ink. During storage, the system stands still and the main force acting on the system is gravity. The only way to prevent particles from sinking is by assuring that a three-dimensional physical network is present in the system during storage.
The physical network must be strong enough to give the material a so-called yield stress, also called yield value or yield point. When a material has a yield stress, it implies that during storage, at low shear stress, the material behaves as an elastic solid. This implies that the material has an infinite viscosity during storage. The solid particles are ‘frozen-in’ and sedimentation is prevented.
Rheology additives
A physical network can be created in a paint or ink by using a suitable rheology additive, often referred to as anti-settling agent. The additive is used to prevent sedimentation and settling. The physical network has to be strong enough to resist the gravitational force during storage. On the other hand, the network must be weak enough to be broken down as soon as sufficient shear force is applied. Many rheology additives are developed for a wide variety of paints and inks.
A few examples of rheology additives, used to prevent sedimentation:
- Fumed silica consists of small solid particles that build a physical network in a material at low shear stress. Many fumed silicas are available, including Aerosil® products from Evonik.
- Certain solid particles with a platelet shape can form a house-of-cards structure in a paint during storage. Well-known are the smectite clays, like the Bentone® products from Elementis.
- Special polymers, designed for the purpose, can also build a physical network in a paint or ink, for example: polyurethanes, polyacrylates, modified urea’s and cellulose ethers.
Need help researching rheology additives?
Prospector can help speed along your research with technical datasheets and access to global suppliers. Get access to free samples today!
Create your free account today!
Bio-based materials
The interest in bio-based materials for paints and inks increases. Two bio-based additives that are used to prevent sedimentation during storage of waterbased paints are of special interest:
- Xanthan gum is a high molecular weight polysaccharide that is fully water soluble. A commercial xanthan gum for waterbased paints is Vanzan® from Vanderbilt Minerals.
- Microfibrillated cellulose (MFC) consists of a three-dimensional network of natural cellulose fibrils1. MFC does not dissolve in water. The cellulose fibrils are present as finely divided solid cellulose branches suspended in water. The physical network is obtained via entanglement of the fibrils, in combination with hydrogen bonding, thus giving a high yield stress in water-based paints. A commercially available MFC product is Exilva® from Borregaard.
Adding less than 1 percent of a rheology additive will be enough to fully prevent sedimentation during storage of a paint or ink, provided that the right additive is selected for the system.
Reference
- Let’s Get Technical: Best Personal Care Formulation Technology of 2017, George Deckner, 19 January, 2018.
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



Thank you. That was interesting
Interesante, la separación y/o decantación es de lo mas delicado en pinturas, difícil de manejar, hay aditivos muy buenos de Elementis, Basf, etc.
Interesting, separation and / or decanting is the most delicate in paintings, difficult to handle, there are very good additives from Elementis, Basf, etc.
Thank you doctor Beetsma.
Can you tell me some analytical methodologies to predict sedimentation in paints using a rotational rheometer?
It is difficult to do a good quantification of the yield stress of a paint by using a simple rotational rheometer. Starting point is to, at least, use a rheometer that can start with zero shear stress and then slowly increase the shear stress and detect when flow in the paint starts. This gives a rough indication of the yield stress of the paint.
Best is to use a scientific rheometer and do so-called oscillatory tests at constant frequency.
I advise to find out what is possible with your rheometer. If you are not sure about this: contact the supplier of the rheometer.
Dr Beetsma i like to know How effective are the anti-flocculant dispersants to avoid sedimentation and how they work?
Dispersants are designed to prevent flocculation (= the spontaneous gluing together of solid particles caused by attractive forces between the particles). These additives do their job because the molecules adsorb at the surface of the particles and they provide repulsive forces between the particles. In the Knowledge Center on the UL Prospector website you can find some articles about dispersants.
Sedimentation is NOT prevented when only dispersants are used. Mostly, sedimentation proceeds FASTER when the right dispersant is used in the right amount!
If a liquid formulation contains particles of high density, a rheology additive is needed to prevent sedimentation.
So: in general a combination of dispersant and rheology additive is needed to prevent both flocculation and sedimentation.
it is very good technical literature for begining of water-based paint R&D.
@Alvaro Hernandez ,
Thanks for referring BASF’s position in supplying formulation additives to prevent particles flocculation. BASF’s rheology modifiers include Alkali swellable emulsions (ASE), Hydrophobically modified alkali swellable emulsions (HASE), Hydrophobically modified polyurethanes (HEUR), Hydrophobically modified polyethers (HMPE), Attapulgites, and Castor oil based thickeners.
http://www.basf.com/formulation-additives Follow us on Twitter @BASF_FoAd
why Mostly, sedimentation proceeds FASTER when the right dispersant is used in the right amount!
A true dispersant is solely used to prevent flocculation of solid particles in a liquid. When the right dispersant is used in the right amount, so-called ‘free-flowing particles’ in liquid result. The particles do not interfere with each other until they come close to each other. Then they repel each other, thus preventing flocculation. When the solid particles have a density that is higher than the density of the surrounding liquid, the particles sink because of gravity (= sedimentation). The particles stack upon each other on the bottom of the can. The repulsive forces between the particles are strong enough to prevent gluing together of the first particles that arrive at the bottom. However, the compressing force becomes too strong when more-and-more particles stack upon each other. Then settling results: the particles glue together on the bottom of the can forming a solid or rubbery layer. The better the dispersant is, the more ‘free-flowing’ the particles are and the faster sedimentation and settling proceeds. Also, the better the dispersant the more solid the sediment will be.
Thank you doctor Beetsma.
This article is very useful for me. I d like to know It is possible to use CAB to prevent sedimentation. I have delamination of resin with CAB, is it problem in resin or we might be solve this problem?
Dear Vladysav,
Thanks for your positive reply. Mostly, CAB is not used to prevent sedimentation. I presume that with ‘delamination’ you mean that you have a problem with adhesion. I advise to check adhesion of your system as naked as possible. This implies: only resin(s) and solvents and/or water. This naked system should have an excellent adhesion. If not, then you must find out why adhesion is not good. See for example the article Achieving Superior Adhesion of Coatings on UL Prospector.
Greetings, Jochum Beetsma.