
Companies in coating and ink technology try to lower the VOC (Volatile Organic Compound) content of their systems. The use of powder coatings, water-based systems and UV-curable coatings and inks has increased substantially over the past decades. Another option to reduce the emission of organic solvents is to increase the solids content of solvent-based systems. This article describes a few concepts that are used to adjust the resin system in order to lower the viscosity so that less solvent is needed to bring the paint or ink to application viscosity.
Need to reduce the VOCs in your paint formulation?
Prospector has hundreds of low VOC material listings with technical data and the ability to request samples.
Get material data
Branching
A key effect causing high viscosity of resin solutions is that resin molecules of moderate to high molecular weight have long tails that stretch out. This results in chain entanglement: the tails hinder each other just like pieces of spaghetti do. An approach to reduce chain entanglement is to design resin molecules that have a more spherical shape. Such polymeric molecules are more branched.
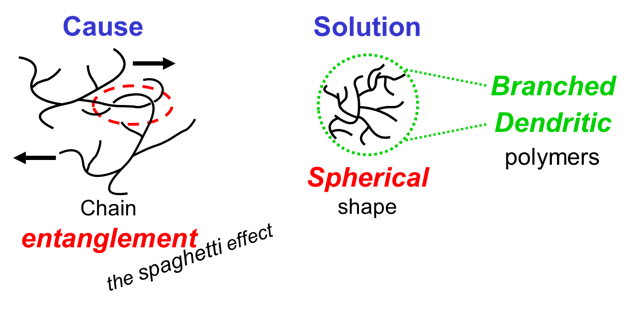
Resin molecules that have a higher degree of branching compared to conventional resins are called hyperbranched resins or dendrimers. Such polymers have lower resin viscosity. Therefore, less solvent is needed to obtain application viscosity of the complete system.
Several innovative companies, like DSM Coating Resins and Perstorp, are strong in (raw materials for) hyperbranched resins. Boltorn™ H2004, for example, is a hyperbranched hydroxyl-functional polymer. Each molecule contains six reactive primary hydroxyl (-OH) groups. Hyperbranched polymers like H2004 can, amongst others, be used as a building block in the synthesis of resins for high-solids systems.
Chainstopper solvents
Intermolecular interactions between different resin molecules create a physical network, which is another phenomenon causing high viscosity of resins that are dissolved in organic solvents. Physical networks that may strongly contribute to viscosity can be temporarily broken down when the interacting groups are blocked, for example by using suitable chainstopper solvents. The solvents evaporate during film formation and the physical network can build up again, thus contributing to physical drying and mechanical properties.
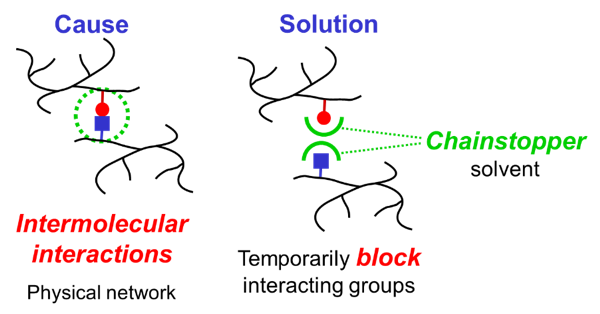
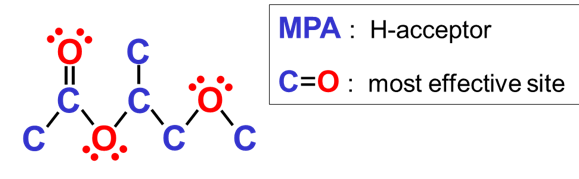
Most physical networks in paints and inks are based on hydrogen bonding. A useful approach to lower viscosity is therefore to use solvents that are able to form one hydrogen bond with a resin molecule, thus ‘stopping’ the physical network. Solvents that contain a C=O group are able to substantially reduce the viscosity of systems based on alkyds and polyesters. Such solvents can act as hydrogen acceptor. MPA, 1-methoxy-2-propyl acetate, is a solvent that is suitable to lower the solvent content of paints based on polyester resins and alkyd resins.
Reactive diluents
The viscosity of binder systems can be lowered by combining polymeric resins with reactive diluents. ‘Reactive’ means that the molecules join in the crosslink reaction during film formation. This implies that the molecules must contain reactive groups of the same sort as the resin molecules. The word ‘diluent’ implies that the molecules must be miscible with, and lower the viscosity of, the resin system. Reactive diluents must have a specific chemical structure as well as a low molecular weight in order to comply with these two criteria.
Reactive diluents must not contribute to VOC, implying that a good reactive diluent does not evaporate during film formation. Reactive diluents are widely used in, for example, 2-component epoxy-amine systems. Epoxy-functional molecules of low molecular weight are combined with conventional epoxy resins to lower the viscosity of the complete binder system.
The concept of combining resins and reactive diluents to lower the viscosity of a resin system can be used in UV-curable paints and inks as well. Rahn product Miramer M210 is an example of a di-functional unsaturated reactive diluent for UV systems.
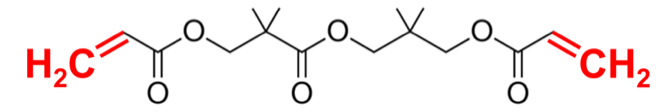
The low viscosity of M210 delivers a strong diluting effect. The two reactive groups of the molecule (C=C) participate in the crosslinking of the binder system during film formation.
Further reading:
- Volatile Organic Compounds: A Brief Regulatory Overview of VOCs
- Rheology of waterborne paints
- Improving Performance with Monomers and Oligomers
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



Thanks for your article. It is very interesting for us as a producer of resins for coatings and thermosetting plastics. I would like to have closer relationship with you here in Andercol Colombia SA.
Also we would like to have some samples of Boltom H2004, Miramer M210 and MPA
Hi Alberto,
You will need to contact the suppliers for samples:
https://www.perstorp.com/en/products/boltorn_h2004
https://www.ulprospector.com/en/na/Coatings/Detail/458/32772/MIRAMER-M210?st=31
https://www.ulprospector.com/en/na/Coatings/search?k=mpa&st=31
Hope this helps!
Angie
Content Manager, Prospector Knowledge Center