 Key concepts
Key concepts
- The thickener largely determines the grease properties.
- Soap is the primary thickener for 90 percent of all greases.
- Simple soaps have a metallic base with a single fatty acid; complex soaps have a metallic base and multiple fatty acids as complexing agents.
- These soaps self-assemble into micelles that allow a compound that is normally insoluble, such as soap, to dissolve.
Grease was first used on chariot axles more than 3,000 years ago. Today more than 80 percent of bearings are lubricated with grease. Lithium soap greases, the most prevalent, were introduced in the early 1940s. Lithium complex greases, introduced in the 1960s, are becoming the most prevalent in North America.
A soap is, by definition, a metal salt of a fatty acid. The National Lubricating Grease Institute defines grease as, “a solid to semi-solid product of dispersion of a thickening agent in a liquid lubricant. Additives imparting special properties may be included (1).”
Grease making is a relatively simple time-temperature process: a one-pot batch method. For a soap grease, fatty acids are added; if it is non-soap, the other constituents are put into base oil. Common acids include the high-molecular-weight fatty acids, stearic acid and 12-hydroxystearic acid and short-chain complexing acids such as tallow, azelaic acid and sebacic acid. Once the acid gets up to temperature (i.e., the fatty acid melts) the metal base is added. The process is called saponification or soap making. So basically acid + base = soap + water (see Figure 1).
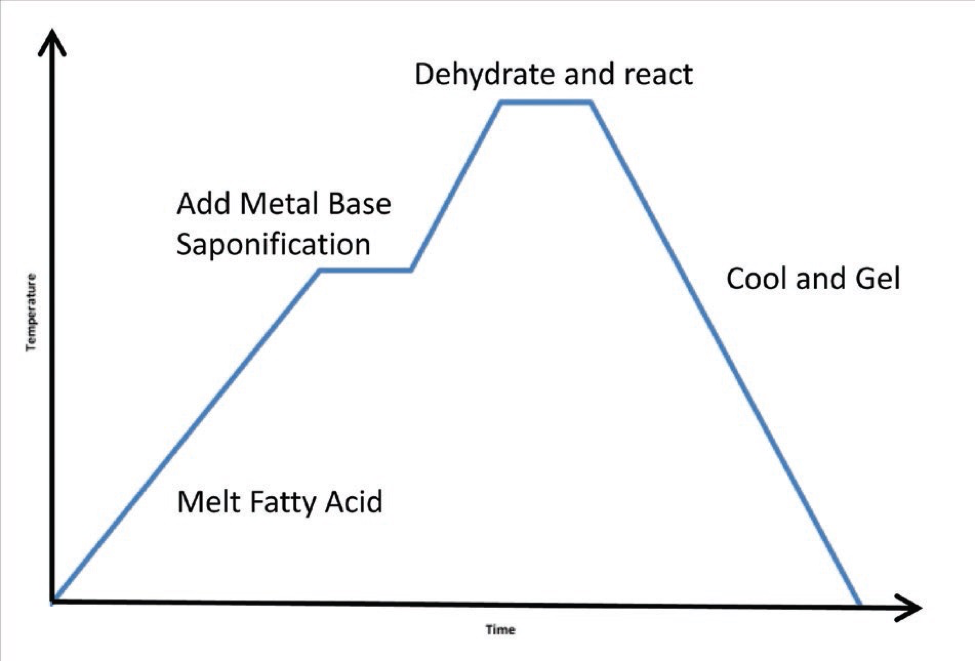
Then, because there needs to be very little water in lubricants, all the water is removed. Once that is done, the material is cooled and gelled—this is the point where the mixture becomes a grease. Next the mixture is adjusted for consistency by adding base oil (additives might be added, as well). It may have to be reheated, recooled and tested several times to get the consistency that is required for the product. Most people think grease is primarily thickener, but in actuality it is mostly oil. Soap concentration in oil is typically 10-20 percent.
Types of thickeners
The thickener defines the type of grease. There are three or four different types of materials that go into thickeners. The focus in this article is on organic thickeners such as lithium stearate, sodium dodecylsulfate and diurea. There are simple greases and complex greases, depending on the types of fatty acids used.
- Simple soaps. The main thickener used in grease is a metallic soap. These metals include lithium, aluminum, sodium and calcium.
- Complex soaps. Greases with complex soap thickeners are becoming more popular because of higher operating temperature and superior load-carrying abilities. Complex greases are made by combining the metallic soap with a complexing agent. The most widely used complex grease is lithium based, made with a conventional lithium soap and low-molecular-weight organic acid as the complexing agent.
- Non-soaps. Non-soap thickeners make sense in special applications such as bentonite clay for high temperatures where it does not melt (2).
Common thickeners include:
Soaps (comprising about 90 percent of all greases used)
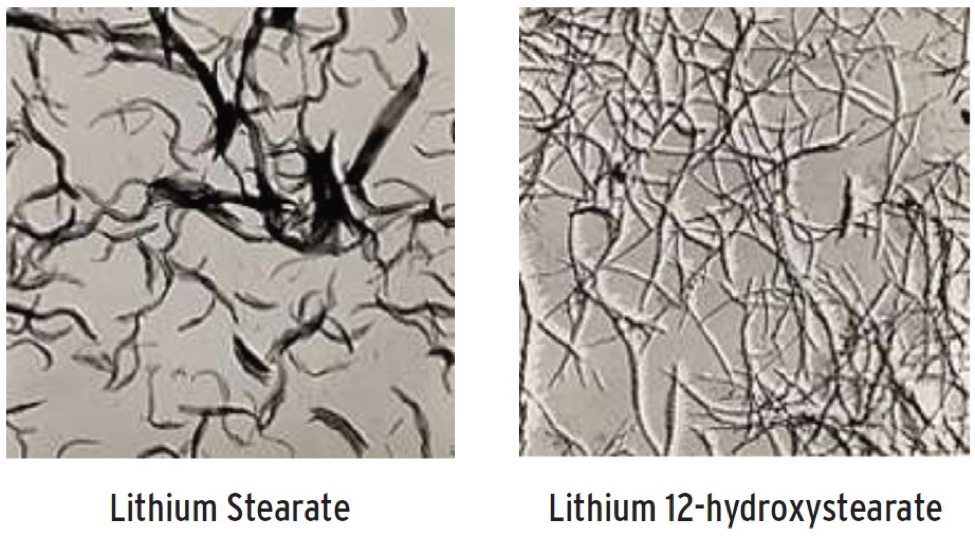
- Lithium: Because lithium soaps are very efficient thickeners, lithium 12-hydroxystearate greases are the most prevalent. Lithium greases provide good lubricity and have great shear stability, thermal resistance and relatively low oil separation. Antioxidants are added to improve oxidative resistance (see Figure 2).
- Calcium: These greases have better water resistance than lithium greases. They also have good shear stability. However, they have low-dropping points, do not have good operating temperature range and can only be used in operating conditions up to 110 C (230 F).
- Sodium: These greases offer high-operating temperature, up to 175 C (347 F) but are confined to operating conditions no higher than 120 C (248 F) because of poor oxidative stability and high oil bleed. They also are not very water resistant. However, they do provide good lubricity and shear stability.
- Aluminum: These have excellent oxidative resistance and good water resistance. But they have a low-dropping point of only 110-115 C (230-239 F). Their usage is generally limited to operating conditions less than 80 C (176 F). When these greases overheat in bearings, they cause sharp torque increases.
Learn more about grease chemistry and micelle formation
This excerpt is based on a webinar originally presented by Paul Shiller for the Society of Tribologists and Lubrication Engineers (STLE) Education. You can read the full summary article at stle.org, as well as access the webinar, Grease Chemistry: Thickener Structure.
References
- Efficient Plant/Maintenance Technology: Grease Basics
- Even though the thickener may be able to withstand high temperatures, the base oil will oxidize quickly at elevated temperatures.
About the presenter
 Paul Shiller received his doctorate in physical chemistry from Case Western Reserve University in Cleveland, studying the surface reactions at fuel cell electrodes. He holds a Master of Science degree in chemical engineering also from Case Western Reserve University, where he studied the characteristics of diamond-like films. Shiller holds a Master of Science degree in chemistry studying the spectro-electrochemistry of surface reactions, and he received a Bachelor of Engineering degree in chemical engineering from Youngstown State University.
Paul Shiller received his doctorate in physical chemistry from Case Western Reserve University in Cleveland, studying the surface reactions at fuel cell electrodes. He holds a Master of Science degree in chemical engineering also from Case Western Reserve University, where he studied the characteristics of diamond-like films. Shiller holds a Master of Science degree in chemistry studying the spectro-electrochemistry of surface reactions, and he received a Bachelor of Engineering degree in chemical engineering from Youngstown State University.
Shiller joined The Timken Co. as a product development specialist for lubricants and lubrication in 2004. He then became a tribological specialist with the Tribology Fundamentals Group at the Timken Technology Center in North Canton, Ohio. In 2011 he moved to the University of Akron in an industrial innovation/collaboration effort as a research scientist. He is currently a research assistant professor at the university working in the Timken Engineered Surfaces Laboratory. You can reach Shiller at paul.shiller@uakron.edu.
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



What could be the possible causes for the thickener structure breaking down after a few months storage eventhough the grease passed all the manufacturing QC checks?
Why do we charge boric acid after lithium in complex grease
I think because of idle on the shelf for quite some time makes the grease start separation.The other possible reason is the choice of acid and base oil plus thickener type.
Why would PTFE mixed in PFPE becomes a gum (rubbery appearance). Any possible explanation.