Introduction
In Part I of this article, expert Jochum Beetsma discussed the definition of PVC, including examples of PVC calculations and the PVC of various types of coatings. In Part II, we will discuss Critical Pigment Volume Concentration (CPVC), pigment oil absorption (OA), and the impact that pigment shape and density have on coatings properties.
Critical Pigment Volume Concentration
As the PVC increases, many properties of a coating change abruptly. These changes occur at the CPVC. CPVC can be defined as the point at which there is just sufficient binder to provide a completely absorbed layer on the pigment surface as well as all the interstitial spaces between the pigment particles in a close-packed system.
Diagram of Paint at CPVC
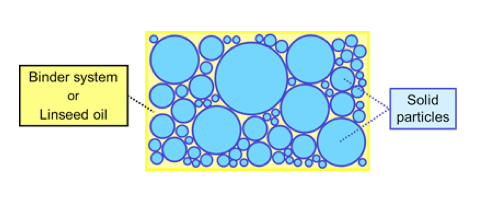 The CPVC for a pigment combination can be calculated from the oil absorption (OA) provided that the OA value is based on a non-flocculated dispersion. OA is expressed as grams of linseed oil per 100 grams of pigment.
The CPVC for a pigment combination can be calculated from the oil absorption (OA) provided that the OA value is based on a non-flocculated dispersion. OA is expressed as grams of linseed oil per 100 grams of pigment.  ρ is the density of the pigment(s), and 93.5 is 100 times the density of linseed oil (EU). Both OA and CPVC are expressed as percentages and not as fractions. The definitions of both OA and CPVC are based on close-packed pigment-binder with just sufficient binder to absorb at the pigment’s surface and fill all the interstices between the pigment particles. An example of the calculation of CPVC of a white alkyd (EU) finish using rutile titanium dioxide (EU) with an oil absorption value of 20 (# of grams of linseed oil/100 grams of pigment) and a pigment density of 4.2 g/cc follows:
ρ is the density of the pigment(s), and 93.5 is 100 times the density of linseed oil (EU). Both OA and CPVC are expressed as percentages and not as fractions. The definitions of both OA and CPVC are based on close-packed pigment-binder with just sufficient binder to absorb at the pigment’s surface and fill all the interstices between the pigment particles. An example of the calculation of CPVC of a white alkyd (EU) finish using rutile titanium dioxide (EU) with an oil absorption value of 20 (# of grams of linseed oil/100 grams of pigment) and a pigment density of 4.2 g/cc follows: 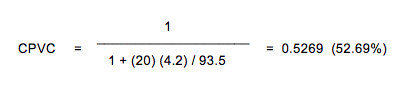 As the pigment density and/or the OA increases, the CPVC decreases. Above the CPVC, air voids are present (film density decreases) and below the CPVC, the pigment particles are separated. The dramatic and abrupt change in the behavior of paint that occurs when passing through the CPVC can be used to determine the CPVC. The abrupt changes in properties include: physical (adhesion, tensile strength/elongation and paint density), durability (resistance to moisture, rust, moisture penetration, blistering, wet adhesion, stain resistance), and appearance (hiding, gloss, tint strength). Other factors that effect water and oxygen permeation include particle shape and particle size. Pigment particles vary in size and shape. Some of the terms used to describe pigment particle structure in increasing order that they depart from sphericity to an increasing degree are as follows:
As the pigment density and/or the OA increases, the CPVC decreases. Above the CPVC, air voids are present (film density decreases) and below the CPVC, the pigment particles are separated. The dramatic and abrupt change in the behavior of paint that occurs when passing through the CPVC can be used to determine the CPVC. The abrupt changes in properties include: physical (adhesion, tensile strength/elongation and paint density), durability (resistance to moisture, rust, moisture penetration, blistering, wet adhesion, stain resistance), and appearance (hiding, gloss, tint strength). Other factors that effect water and oxygen permeation include particle shape and particle size. Pigment particles vary in size and shape. Some of the terms used to describe pigment particle structure in increasing order that they depart from sphericity to an increasing degree are as follows: 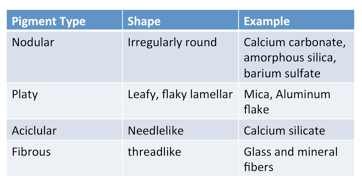 Pigments with platelet shaped particles can reduce permeability especially if they are aligned parallel to the coating surface. Mica (EU), micaceous iron oxide (EU) and metal flakes (EU) are a few examples of such pigments. The smaller the average pigment particle size, the more resistant pigments are to dense packing. For example, the dense packing factor for fine (precipitated) calcium carbonate (EU) is on the order of half that of coarser calcium carbonate. The surface area of a unit weight of pigment varies inversely with the particle diameter. This relationship is especially true for most pigment particles that do not vary greatly in shape from a sphere, nodule or rectangle. Thus, for a given weight of pigment particles, halving the diameter doubles the surface area, and the greater the surface area for a given pigment, the greater the vehicle demands.
Pigments with platelet shaped particles can reduce permeability especially if they are aligned parallel to the coating surface. Mica (EU), micaceous iron oxide (EU) and metal flakes (EU) are a few examples of such pigments. The smaller the average pigment particle size, the more resistant pigments are to dense packing. For example, the dense packing factor for fine (precipitated) calcium carbonate (EU) is on the order of half that of coarser calcium carbonate. The surface area of a unit weight of pigment varies inversely with the particle diameter. This relationship is especially true for most pigment particles that do not vary greatly in shape from a sphere, nodule or rectangle. Thus, for a given weight of pigment particles, halving the diameter doubles the surface area, and the greater the surface area for a given pigment, the greater the vehicle demands.
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



What if it is a mixture of Titanium and Aluminium paste? how to calculate OA since Aluminium does not have an OA value?
Dir sir
thank you for this article, it is very useful for the producers and chemists, I have a question for oil absorption. how I know that the pigment absorbs the oil and stopped at the limited point that no absorb above it, by notice or something else ??
Good post shared.