 Whenever soft matter (Tg < room temperature) like alkyds are emulsified, stable emulsions can be prepared most of the time, either via a low interfacial tension or a high-shear process.
Whenever soft matter (Tg < room temperature) like alkyds are emulsified, stable emulsions can be prepared most of the time, either via a low interfacial tension or a high-shear process.
However, one can never avoid collisions between particles. In contrast to hard particles (Tg > room temperature), collisions between soft particles will lead to coalescence, unless an excess of repulsive forces are provided. These repulsive forces can be either electrostatic (mostly anionic) or steric (mostly referred to as nonionic). A billiard table offers the best comparative example of hard particles colliding.1
This problem occurs when the boundary layer between two liquids is diffuse or badly defined, and the hydrophilic/lipophilic behavior is undefined. Some emulsified materials like alkyd resins have a very shady definition. It comes as no surprise that the most hydrophobic molecules will prefer to reside in the core, whereas the less hydrophobic molecules prefer the shell.
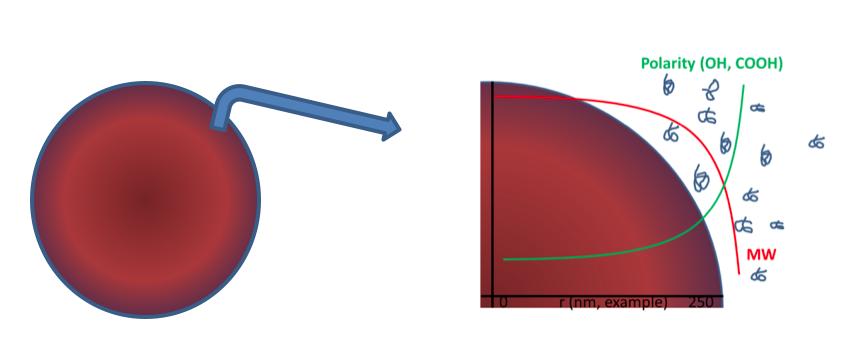
Let’s interpret the above figure from an alkyd perspective.
- In the core (red), there are molecules with a high molecular weight but low hydrophilicity: the ultimate performers.
- In the shell (purple to blue, let’s call it a “fuzzy” layer for the time being), we find molecules with a lower molecular weight but higher hydrophilicity like OH- and COOH values: emulsification aids.
- Surrounding the shell, in the aqueous phase, are molecules with a very high hydrophilicity and a very low molecular weight, like Pentaerythritol with one or two fatty acids attached to it, or two di-acids with one Penta and one fatty acid attached to it. These oligomers are well-known in the polycondensate business and presumably after emulsification, they will assist in a Newtonic behavior of the alkyd emulsion.
Let’s concentrate on this fuzzy layer.
Stabilization mechanisms like the well-known DLVO theory2 will not obey the rules that usually apply to well-defined and even soft interfaces like those obtained with emulsion polymerization. No simple Hamaker constant, no clear zeta potential, just because there is no well-defined interface. Just a fuzzy layer.
This implies that the ideal surfactant will be one that fits this fuzzy behavior, i.e. there should be an amphiphilic block in-between the hydrophobic part and the hydrophilic moiety. Whatever this amphiphilic block is, its length will be determined by how well defined the alkyd is, and thus, among others, by its oil length. An educated guess would be something like 50 nm.
This need for a fuzzy block probably also explains why Sodium Dodecyl Benzene Sulphonate (SDBS), Sodium Lauryl Sulphate and salts of Colophonium are bad surfactants for alkyds.
Coating additives at your fingertips
Don't spend hours searching multiple websites - quickly find technical data on additives like surfactants in Prospector®. You can even order samples and contact global suppliers directly.
Named after scientists Boris Derjaguin, Lev Landau, Evert Verwey and Theodoor Overbeek, the DLVO theory describes interactive forces between two particles as a function of their distance.
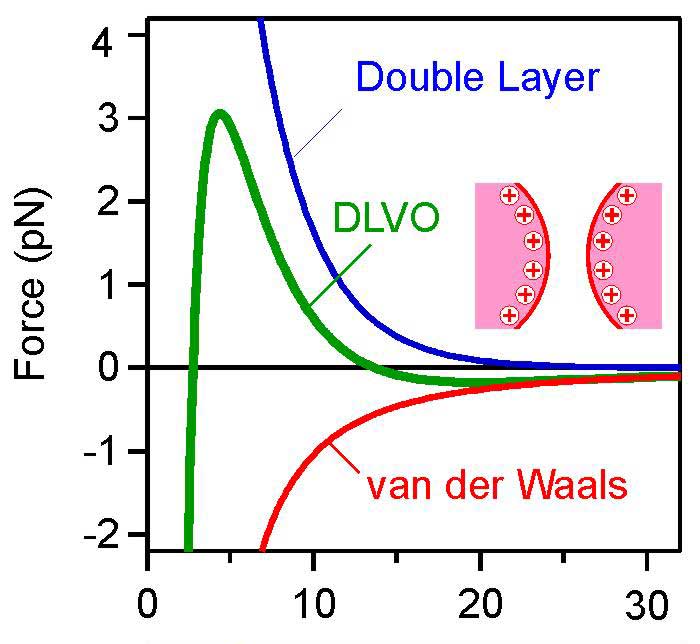
However, with emulsions of soft and less well-defined materials, this is hardly ever the case. DLVO theory applies to some extent, but proper interpretation of the horizontal axis is questionable, in this case the interparticle distance in nm.
Competing with the “fuzzy surfactant,” there is another possible solution.
Rising in popularity is the phenomenon of Pickering stabilization3: make the alkyd particle behave like the aforementioned billiard ball. In this solution, the “fuzzy layer” is covered by, or even filled up by, very hard material. In spite of the same frequency of collisions as can be expected with traditional Brownian motion, there is no risk of coalescence and a stable alkyd emulsion can be obtained. Under the trade name of Secoia, the French binder producer Ecoat has developed an alkyd emulsion using this technology.4
With an increase in the demand for sustainable and preferably water-based coating systems, in a bio-based economy, emulsified alkyds will likely gain in popularity. Traditionally active in the field of water-based acrylics or solvent-based alkyds, large companies like DOW, Arkema, Reichhold and aforementioned Ecoat, have already shown their interest.
Further reading:
References:
- YouTube: Billiards Demonstration
- University of Geneva: Overview of DLVO theory
- The Phenomenon of Pickering Stabilization: A Basic Introduction
- Ecoat Products
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



Hi Ad,
What a super article! Thanks for publishing it and well done.
Peter
Thanks Peter