Performance and Characterization of Non-ionic Surfactant-Activated Microgels in Soap-based Cleansing
Paid content by Lubrizol Advanced Materials
By Brian Figura, Robert Jacobs, Dongcui Li, and Krishnan Chari
Introduction
Alkali-swellable emulsions (ASE) are widely used to modulate the rheology of personal care formulations. However, these polyelectrolyte microgels may be challenged in high pH and high ionic strength formulations. Here we demonstrate the performance of non-ionic surfactant-activated microgels in such a system: a clear, suspending, soap-based cleansing formulation. These polymers, commercially available as the Carbopol® SMART polymers, are cross-linked non-ionic microgels that make use of surfactant-mediated swelling and interaction to provide rheological performance that is independent of pH [1]. We further characterize this system using rheology to gain insight into the structure of the formula. Finally, we show that surfactant-activated microgel technology offers the potential to deliver irritation mitigation and a differentiated sensorial experience.
Results and Discussion
The non-ionic and non-polyelectrolyte nature of the SMART technology is contrasted versus ASE technology in Figure 1. This shows the effect of pH on performance properties for formulations containing 2.5 wt% polymer in a surfactant chassis containing 12 wt% SLES-2, 2 wt% CAPB, and 0.1 wt% sodium chloride. As can be seen from Figure 1, with an ASE polymer, formulation clarity and suspension strength vary significantly as a function of pH, unlike the behavior of the SMART polymers.

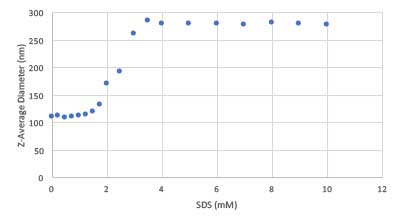
The surfactant association driving this performance is illustrated in Figure 2. Here we show dynamic light scattering (DLS) measurements of the diameter of a SMART polymer in a dilute suspension as a function of increasing surfactant concentration. The details of this, and other methods, have been reported previously [2]. Once the surfactant concentration exceeds the critical aggregation concentration (CAC) (independently determined by proton nuclear magnetic resonance (NMR) to be 1.3 mM for sodium dodecyl sulfate (SDS)), the surfactant begins to associate with the SMART polymer thereby driving the swelling of the microgel [1].
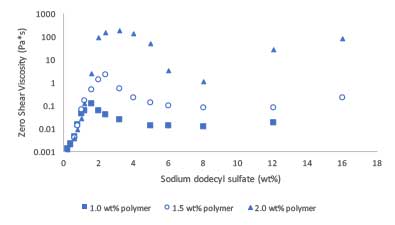
This surfactant response can also be seen in Figure 3. Here, we show zero-shear viscosity as a function of both surfactant and polymer concentration. The peak observed in viscosity as a function of surfactant concentration at polymer concentration above the overlap concentration is indicative of surfactant mediated interaction of the swollen microgels [3, 4]. At the higher concentrations of surfactant, another upturn is observed due to the surfactant morphology changing from spherical micelles to worm-like micelles.
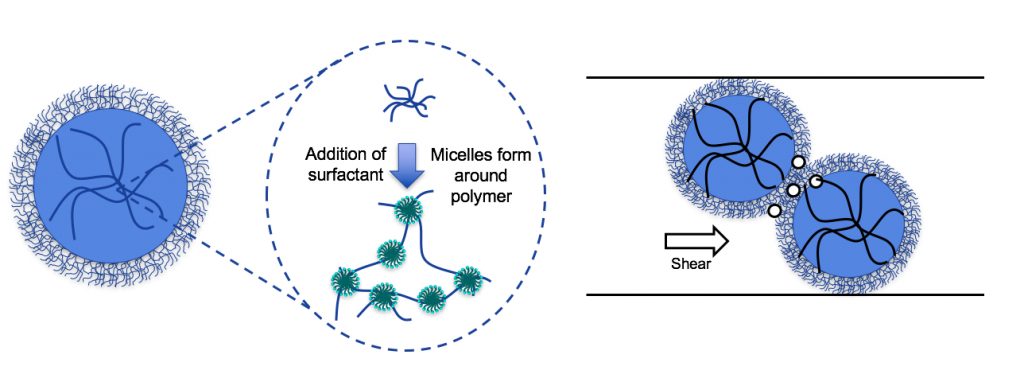
It is important to note that there are two separate modes of surfactant interaction; the swelling of individual microgels by surfactant micelles above the CAC and the micelle mediated interaction of the individual swollen microgels at concentrations above the polymer overlap concentration which creates a network that resists shear and provides a yield stress. This is illustrated in Figure 4.
To demonstrate the utility of this technology in high pH, high ionic strength formulations, in Table 1 we show a representative soap-based cleansing formula using a commercial SMART polymer, Carbopol® SMART 1000. In traditional soap or soap/syndet cleansing formulations, it may be challenging to achieve formulas that deliver high clarity, suspension strength, and desirable aesthetics. The flexibility of the Carbopol® SMART 1000 polymer enables the delivery of these properties in a formulation that remains clear and stable, even under challenging conditions, i.e. low and high temperatures. The properties of this formulation are shown in Table 2.
|
Table 1: Sample Formulation |
|
| Ingredient | Active Concentration (wt%) |
| Lauric Acid | 12.00 |
| Myristic Acid | 2.50 |
| Palmitic Acid | 0.50 |
| SLES-1 | 4.00 |
| CAPB | 3.00 |
| NaCl | 1.50 |
| Na4EDTA | 0.05 |
| KOH | To pH 9.5 – 10 |
| Carbopol® Smart 1000 Polymer | 2.50 |
| DI Water | q.s. to 100% |
| Table 2: Formula Properties | |
| Parameter | Result |
| Viscosity | 5,000 – 7,000 cP |
| Turbidity | 5 – 15 NTU |
| Yield Stress | 3.00 – 4.00 Pa |
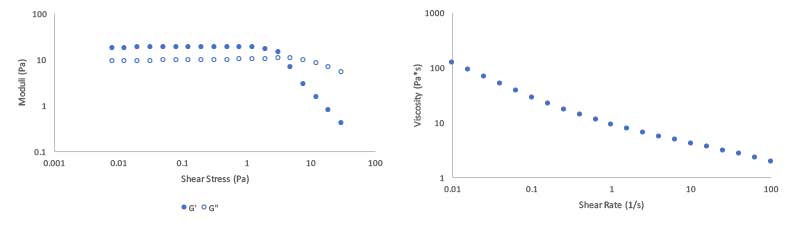
The rheological performance of the polymer in this system is shown in Figures 5a and 5b. In Figure 5a, we show the storage and loss moduli (G’ and G” respectively) as a function of increasing oscillatory shear stress. At low stresses, the polymer microgels respond elastically and exhibit solid-like behavior, enabling the suspension of encapsulates or exfoliating agents. At higher stresses, the material yields (as G’ approaches G”), and the material begins to behave like a liquid, providing aesthetically desirable flow. This flow is shown in more detail in Figure 5b. Here, we examine viscosity of the material as a function of increasing shear rate, and observe a desirable shear-thinning profile.
As we have mentioned, the non-ionic and amphiphilic nature of the Carbopol® SMART polymers enables them to uniquely interact with surfactant molecules, thus enabling clarity and desirable rheological properties. Additionally, the surfactant-polymer interaction enables the Carbopol® SMART polymers to mitigate the irritation caused to the skin by surfactants. A common means of predicting irritation potential is to measure the exposure time of a test formulation required to reduce cell viability to 50 percent of the control viability (ET-50) [5]. Greater irritation potential is linked to lower ET-50 values. To demonstrate this concept, we present, in Table 3, ET-50 values measured on dermal cell cultures for common surfactants: SLS, SLES-1, SLES-2, and SLES-3. As expected, as the surfactant becomes more ethoxylated, the ET-50 increases, and so does the mildness of the solution. We then apply this method to demonstrate the irritation mitigation potential of Carbopol® SMART polymers in the presence of a relatively harsh surfactant, SLS.
| Table 3: Measurement of dermal irritation | |
| Formulation | ET-50 (hours) |
| 3.2 SLS + 0% SMART Polymer | 1.16 |
| 3.2 SLES-1 + 0% SMART Polymer | 3.13 |
| 3.2 SLES-2 + 0% SMART Polymer | 4.31 |
| 3.2 SLES-3 + 0% SMART Polymer | 6.53 |
| 3.2 SLS + 2% SMART Polymer | 2.50 |
The simple addition of the SMART polymer to the surfactant solution results in a 215 percent increase in ET-50, almost approaching the ET-50 value of SLES-1.
In further sensory evaluations, we have performed in-house consumer panel tests to compare formulations made with a traditional ASE polymer to formulations made with a Carbopol® SMART polymer. Formulations made with a Carbopol® SMART polymer were found to give a smoother skin feel during hand washing, followed by a squeakier skin feel on drying, and a smoother powdery feel on total dryness.
Conclusions
We have demonstrated that non-ionic, non-polyelectrolyte surfactant-activated microgel (SMART) technology, represented by the Carbopol® SMART polymers enables pH-independent rheology control in traditional personal care cleansing formulations. This result was then extended to soap-based cleansing formulations. In these soap-based formulations, we have shown that Carbopol® SMART polymers enable consumer-desirable properties such as clarity, suspension, and aesthetic flow. We have further shown that these polymers can mitigate the skin irritation that can arise from surfactant usage, and can yield differentiated sensorial properties from traditional alkali-swellable emulsion technology.
Bibliography
- Chari, K., et al., Surfactant-Activated Microgels: A New Pathway to Rheology Modification. Langmuir, 2013. 29(50): p. 15521-15528.
- Brian Figura, R.J., Dongcui Li, and Krishnan Chari, Performance and Characterization of Non-ionic Surfactant-Activated Microgels in Soap-based Cleansing. Fragrance, 2016. 44(5): p. 54.
- Panmai, S., et al., Interactions between Hydrophobically Modified Polymers and Surfactants: A Fluorescence Study. Langmuir, 2002. 18: p. 3860-3864.
- Piculell, L., et al., Binding of surfactants to hydrophobically modified polymers. Advances in Colloid and Interface Science, 1996. 63: p. 1-21.
- Michael J. Derelanko, C.S.A., Handbook of Toxicology, Third Edition. 2014: CRC Press.
Click Here to view Carbopol® SMART polymer brochure
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



Leave a Reply or Comment