Original article date: July 14, 2017
Updated by Jochum Beetsma – August 2022
This article is the follow-up of article Optical Properties of Pigments: Absorption and Scattering1. Absorption and scattering of light by a pigment in a coating are governed by the chemical composition of the pigment and the size of the pigment particles.
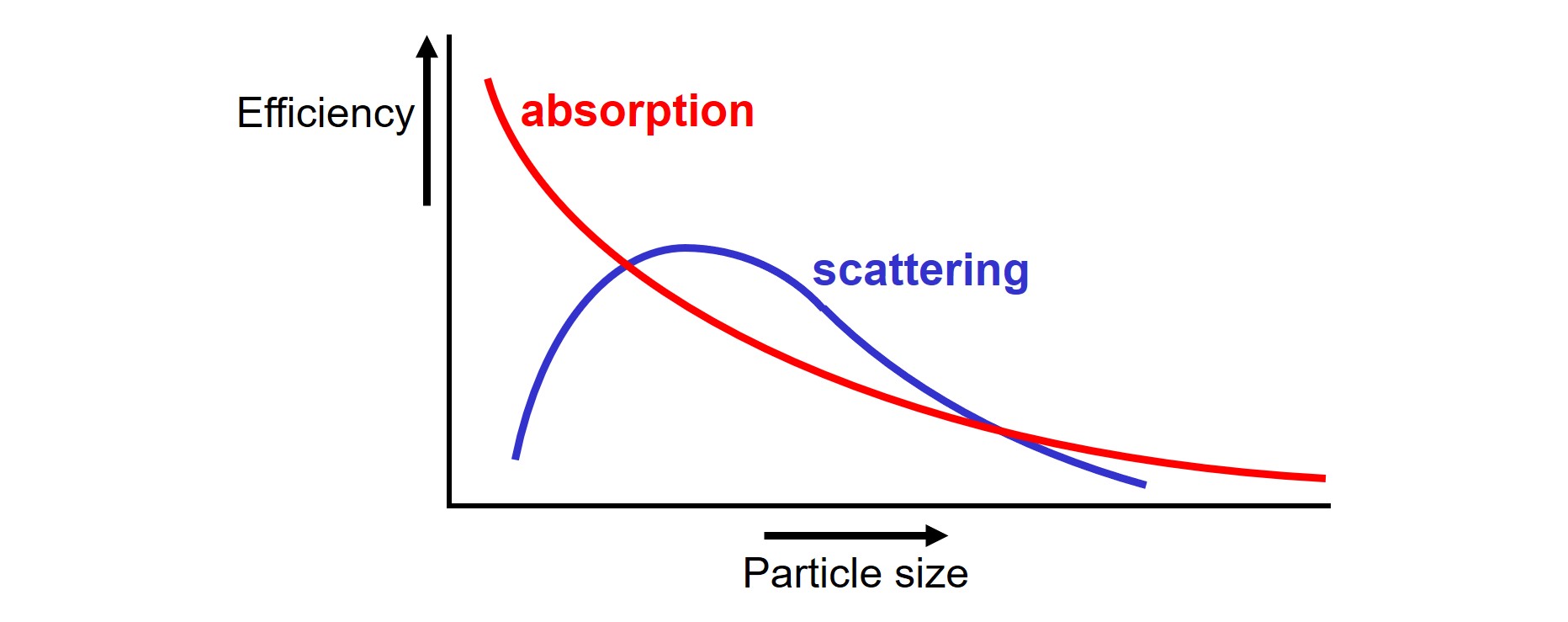
Each molecule in a pigment particle participates in absorption. Molecules in the core of a big particle do not have the ability to absorb light because the light was already absorbed by the molecules that are in the shell of the particle. This implies that the absorption efficiency of solid particles goes up when the particle size goes down: small particles have more surface area per gram of pigment than big particles. The scattering of light by particles depends upon the size of the particles as well. With respect to scattering, there is an optimum with respect to particle size1.
Transparent titanium dioxide
In most coatings, titanium dioxide (TiO2) is used as white pigment giving white color strength as well as hiding power, also called opacity2,3. The underlying principle is two-fold.
First, microscopic TiO2 particles scatter visible light (electromagnetic radiation with wavelength λ ≈ 380 – 750 nm) effectively because TiO2 has a high refractive index, represented by the letter ‘n’. Optimum scattering of visible light by particles in a system is obtained when the particles have a diameter of about half the wavelength of the light. The diameter of opaque TiO2 pigments is in the range of 300 nm for that reason.
Secondly, titanium dioxide does hardly absorb visible light. That is why we see the particles as white.
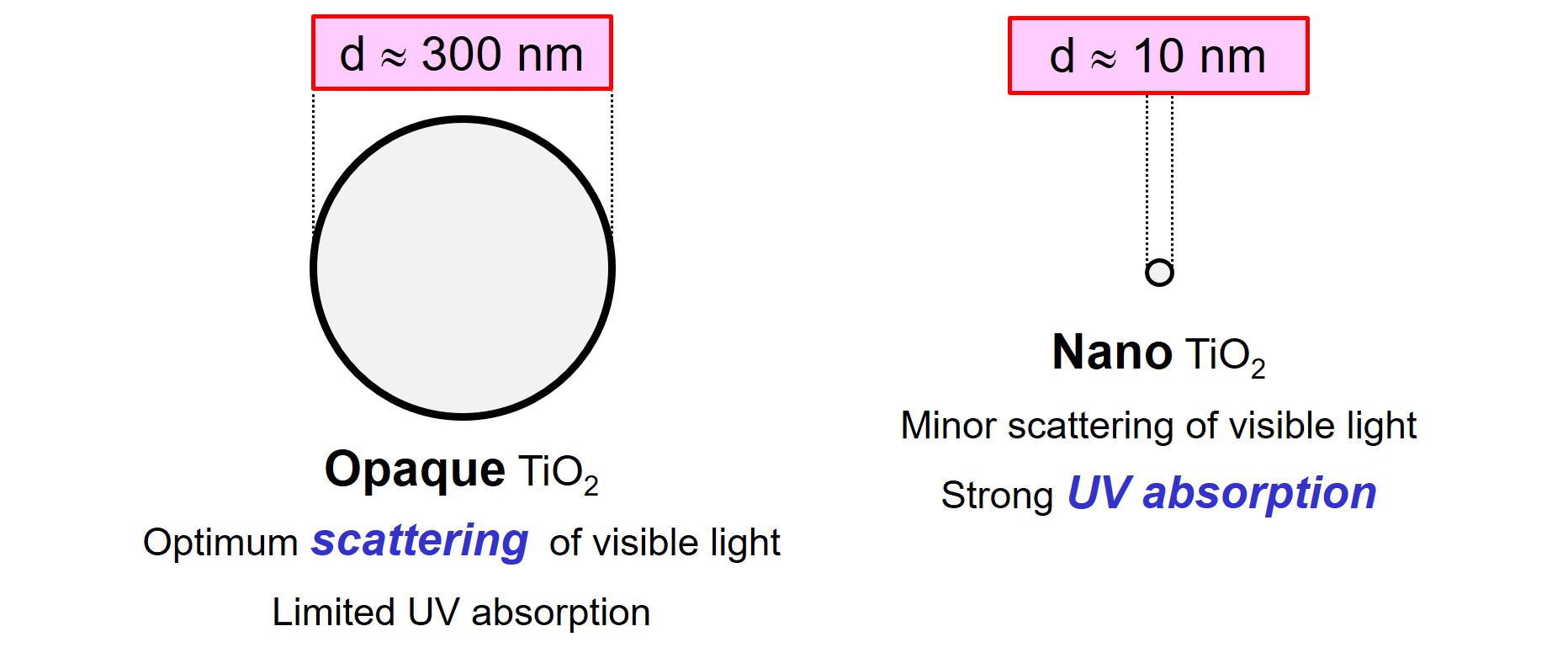
TiO2 pigment absorbs UV radiation effectively, especially when the particles are small. Small particles have a high specific surface area and therefore contain many surface molecules that are most effective in absorbing light. Nano-sized titanium dioxide (nano-TiO2) pigments, with a particle diameter ‘d’ far below 100 nm, are used as a UV absorber in systems like paints, inks and plastics. A coating that contains nano-TiO2 is transparent because the particles hardly scatter visible light.
As nanoparticles are difficult to disperse in a liquid, many companies choose to buy ready-to-use dispersions of nano-TiO2. An example of such a product is CCR 150 of Cinkarna. CCR 150 is a dispersion of 16 weight-% of nano-TiO2 particles, with an average diameter of around 10 nm, in water. The material can be used as a UV absorber in paints, plastics and foils. Because of the small particle size, the product can be used in transparent systems.
Transparent iron oxide pigments
Iron oxide red (Fe2O3, color index PR 101) and iron oxide yellow (FeOOH, color index PY 42) are inorganic solids that are used as color pigments in paints. As these materials have a high refractive index, the particles scatter visible light strongly. Iron oxide pigments are colored, implying that the particles absorb part of the visible light1. For some applications, colored pigments are desired that absorb part of the visible light, but that scatter visible light as little as possible.
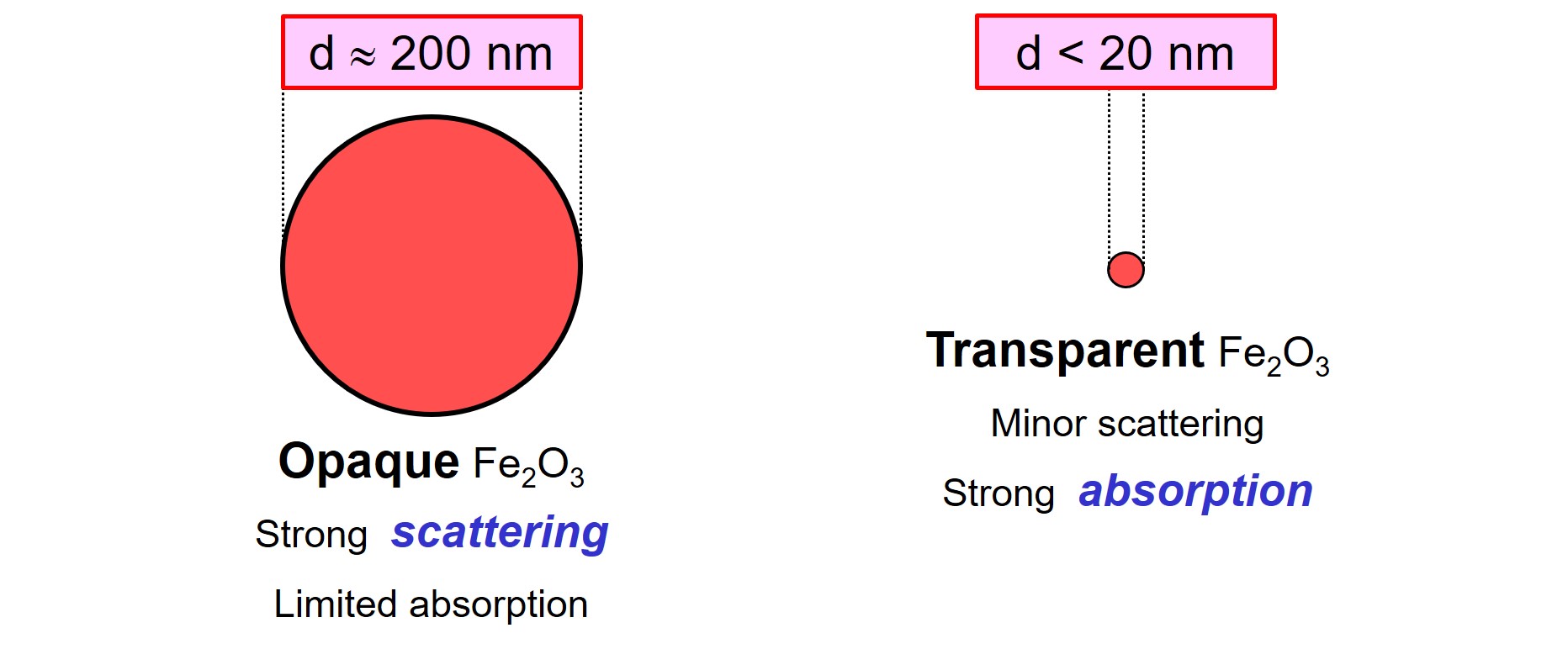
Minor scattering is desired in, for example, transparent wood stains also called wood varnishes. Such coatings must protect the wood and give color to the object. On the other hand, the beautiful wood structure must remain visible. To achieve this combination of properties, the coating must contain pigments that absorb part of the visible light to obtain color, but that hardly scatter visible light to obtain transparency.

An example of a transparent iron oxide red pigment is TRAFe® BTR-201W of Chemsfield. The particle size is not given in the Technical Data Sheet of this product, but the high specific surface area value tells us that the pigment consists of small particles and has a high amount of surface molecules.
TRAFe® BTY-101W is a transparent iron oxide yellow pigment from the same product range.
Transparent iron oxide pigments are difficult to disperse, especially because many of these products consist of needle-shaped particles. For this reason, paint-producing companies often choose to buy ready-to-use pigment concentrates. CLiQ SwissTech supplies dispersions of transparent iron oxide pigments, for example, CLiQTRANS® RW 41 and CLiQTRANS® YW 50.
References
- Article Optical Properties of Pigments: Absorption and Scattering, Jochum Beetsma, 9 March 2022.
- Article Titanium Dioxide: The Misunderstood Pigment, Marc Hirsch, 27 March 2020.
- Article Efficient Use of TiO2 Pigment, Jochum Beetsma, 1 August 2019.
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



excellent article very clear
Very informative
I am really interested in your product, transparent TiO2. I would like to talk to you about it. Please send me email. Thank you!
Dear Junyoung, I do not supply products or raw materials; I am consultant. Please look at the website of UL Prospector to find suppliers + products. For transparent titanium dioxide look, for example, under Cinkarna.
Greetings, Jochum.
Hello Jochum
I have a customer that runs a lot of natural kraft board and they will pick a pantone color out of the pantone book a request a match we match the color pretty colose to the pantone book send a swatch for their aproval but when the colors goes to press it comes out way to light. the colors do contain TiO2 opaque white maybe 33% and the rest clear extender and the dispersion colorants equeling 100% it’s like the white is dominant when it is not? We use a bladed harper hand proofer, with the same volume BCM anilox as the press but yet the press still deliver’s more opacity and doesn’t match the aproved color swatch we get constant complaints? is it becasue the white is heaver in weight per gallon 11.0 opposed 8.5?
Dear Mike,
You mix different colors. I presume that the colorants are dispersions of solid pigment particles in liquid.
I invite you to read the UL article about that: The Basics of Dispersion and Stabilization of Pigments and Fillers.
You description suggests that there are 2 possible problems:
Incomplete separation
When separation (during the dispersion process) is incomplete, further separation can occur during mixing and/or during the application process. When this occurs, the color strength of (one of the) color pigments increases.
Flocculation
When the wrong type (or wrong amount) of dispersant is used, flocculation will occur, resulting in the color strength of the flocculating pigment going down in time.
I cannot judge which of the 2 undesired processes takes place in your case.
Greetings, Jochum Beetsma.