 No matter what surface needs to be painted, performance begins with proper cleaning and surface preparation. This article will concentrate on the essential issues in the manufacturing process necessary to ensure successful metal treatment and resultant coating performance. As there are hundreds of surface treatments, we will address the major factors that influence phosphate metal pretreatment which are one of the most widely used pretreatment chemistries. Phosphate treatments are used on steel, zinc and aluminum substrates.
No matter what surface needs to be painted, performance begins with proper cleaning and surface preparation. This article will concentrate on the essential issues in the manufacturing process necessary to ensure successful metal treatment and resultant coating performance. As there are hundreds of surface treatments, we will address the major factors that influence phosphate metal pretreatment which are one of the most widely used pretreatment chemistries. Phosphate treatments are used on steel, zinc and aluminum substrates.
The pretreatment process for metal surfaces provides multiple benefits as it is the foundation of the paint layering system. A quality metal treatment process enhances adhesion between the metal and paint layers by providing a more uniform surface and provides greater corrosion resistance with less undercutting of the paint film.
Table I – Typical Spray or Immersion Process involved in Phosphate Pretreatment
| Process Step order | Purpose | Chemicals | Potential problem(s) |
|---|---|---|---|
| 1. Cleaning (see Figs I & II) | Remove soils, mill oil, lubricating oil and drawing compounds, dissolution of metal oxide(s), precipitate hard water deposits | Alkaline Cleaner |
|
| 2. Water rinse(s) | Remove residual detergents and deposits | Quality tap water and/or reverse osmosis (R/O) water |
|
| 3. Rinse Conditioner (see Fig III) | For Phosphate- Aids in the development of the proper phosphate crystals on the metal surface | Colloidal Titanium Salts and additives | Destabilization of the Ti Colloid:
|
| 4. Phosphate Step (See Fig IV) | Forms a microcrystalline coating to enhance paint adhesion and corrosion resistance |
|
|
| 5. Rinse | Stops the chemical reaction on the metal surface | Water | Water must be clean |
| 6. Post Rinse | Fill voids in pretreatment | Hexafluorozirconqic acid | Proper control of pH, time , temperature and pressure (spray) |
| 7. Deionized (D.I.) Rinse(s) | Remove any residual chemicals and to provide a clean surface for coating | D.I. Recirculating rinse, followed by a D.I rinse | Carry over of chemicals and other contaminants from previous steps. Must ensure that D.I. water quality is maintained |



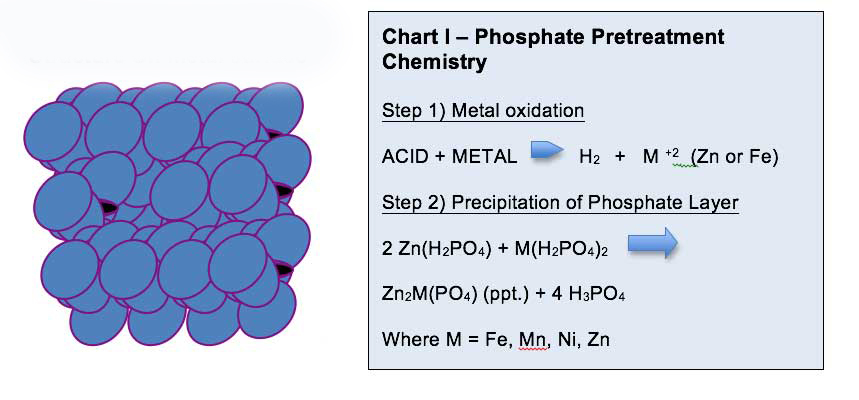
Step 6, the rinse step, is essential to stop the reaction on the metal surface, with the subsequent post rinse utilizing hexafluorozirconic acid to seal the spaces between the Zn Phosphate crystals.
This post-rinse step helps to pacify the metal surface and thus ensure that optimum corrosion resistance is possible. Due to chrome toxicity, hexafluorozirconic acid has replaced chromic acid in many cases to properly seal the gaps in the phosphate crystals.
The ultimate phosphate crystal size, composition and morphology (nodular or crystalline) varies with the substrate selected, and whether the treatment process is spray or immersion.
For example, there are multiple forms of Zinc Phosphate with different chemical compositions and effectiveness when it pertains to corrosion resistance. The forms of Zn phosphate include hopeite, phosphophyllite, phosphonickellite and phosphomangallite, with hopeite offering the least corrosion protection as it is more soluble in an alkaline environment.
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



Many coatings formulators use our 1.5-nanometer phosphato and pyrophosphato titanates and zirconates to promote intrefacial adhesion and form an anti-corrosive atomic monolayer on metal surfaces while dispersing pigments and extenders and improving overall rheology and polymer mechanical properties.
There are hundreds of ACS CAS Abstracts in the literature showing efficacy of phosphorus and heteroatom functional organometallics in energetics, cosmetics, electronics, magnetic recording media, copier toner, powder coatings, proppants, etc. – as well as anti-corrosion and flame retardant compositions.
What metals is this treatment suitable for?
Can u tell me how to make phosphate chemicals?
For consulting support, please contact us through our website – Chemicaldynamics.net.
Thank you,
Ron Lewarchik