Original article date: Oct. 28, 2016
Introduction
Pigments and fillers, both consisting of solid particles, are important raw materials for paints. The powdery materials are wetted, separated and stabilized during paint production1. Separated solid particles in a liquid have the tendency to glue together because of attractive Van der Waals forces between the particles. The spontaneous, and undesired, gluing together of solid particles in a liquid is called flocculation.
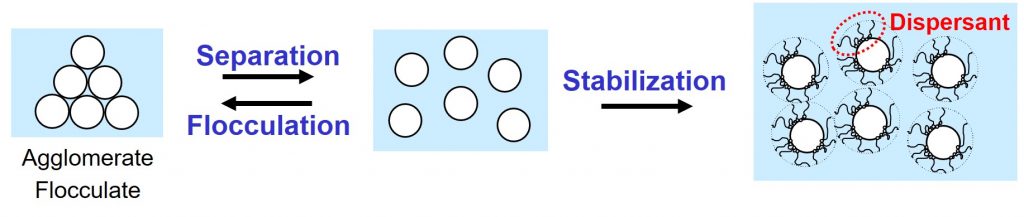
Colloidal stability, the stability against flocculation, can be assured by using the right type of dispersant2,3 in the right amount. A suitable dispersant assures that the solid particles repel each other. The repulsive forces, provided by the dispersant molecules adsorbed at the surface of the solid particles, must be stronger than the attractive Van der Waals forces.
Two mechanisms, steric stabilization and electrostatic stabilization, can be used to prevent flocculation. This article is about steric stabilization.
Steric stabilization
Flocculation is prevented via steric stabilization when all solid particles are covered with polymeric tails that dissolve in the liquid surrounding the particles, the so-called continuous phase. In a water-based paint, for example, the tails must be water-soluble to provide steric stabilization.
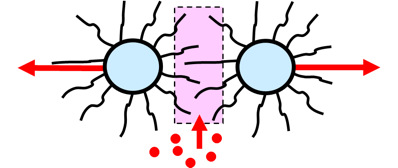
The concentration of soluble tails in the area between particles goes up when the particles approach each other. This is unfavorable from an energy point of view. The system reacts by diluting the tails: solvent molecules (or water molecules in a water-based system) flow to the area between the particles. The result of this osmotic flow is that the particles move away from each other. Mutual repulsion of the particles is governed by the solubility of the polymeric tails in the liquid. The principle of steric stabilization can be used in both solvent-based and water-based systems.
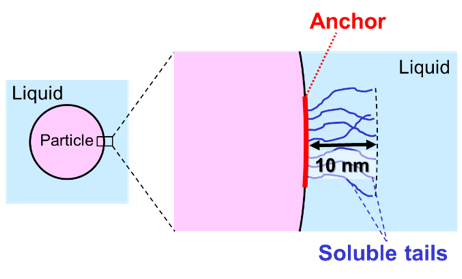
Polymeric dispersants
Dispersants that provide steric stabilization are polymeric molecules consisting of two functional parts. An anchor block must assure that the molecules adsorb at the surface of the particles. Secondly, at least one soluble tail is attached to the anchor block. The tails must have a certain minimum length, and the concentration of tails in the layer around the particles must be high enough to give the desired osmotic effect.
An example
Disperbyk®-190 is a comb-shaped dispersant that can be used in water-based systems to stabilize a wide variety of, both organic and inorganic, pigments and fillers against flocculation. The anchor block of each molecule contains several anchoring groups, also referred to as ‘pigment affinic groups.’ Strong adsorption at the surface of a range of solid particles is obtained in this way. The additive is supplied as a ready-to-use solution of high molecular weight block copolymer in water.

Key aspects of steric stabilization
A relatively high dosage of polymeric dispersant is needed when the additive gives steric stabilization. This is because a substantial percentage of the solid surface of the particles must be covered with polymer. Otherwise, the concentration of soluble tails in the layer around the particles is too low to give the osmotic effect. The dosage of dispersant calculated on the amount of solid surface area of the particles, the dispersant load, is much higher for dispersants that provide steric stabilization than for dispersants that give electrostatic stabilization.
An advantage of steric stabilization, compared to electrostatic stabilization, is that it has a low sensitivity towards changes in pH and the presence of salts. This is important in water-based systems.
It is difficult to give particles an electrostatic charge in solvent-based systems. Because of that, steric stabilization is the main mechanism used in solvent-based systems.
References
- Article The Basics of Dispersion and Stabilization of Pigments and Fillers, Jochum Beetsma, 31 January 2020.
- Article Dispersants – The Understood Misunderstood Additive, Marc Hirsch, 10 September 2021.
- Article Understanding Dispersants, Marc Hirsch, 19 February 2016.
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



antifloculantes y dispersantes para el archivo
does the dispersant will increase the surface energy of the coating. I have tried many surfactants in a low surface energy coatings which contains silica nano particles. But the surface energy increased due to these dispersants and affects the coating performance.
Can you confirm about your product.
Best regards
J Mohan sunder
[email protected]
Bom dia , gostaria de saber se este mesmo principio se aplica em uma dispersão de pigmentos utilizados em tintas de segurança onde o efeito do pigmento é percebido mediante a exposição a uma luz uv e nosso grande problema é a estabilização da sedimentação pois o produto não pode ter uma viscosidade muito elevada pois no processo de aplicação que é feito através de canetas em tubos muito finos eque conduzem a tinta até o bico aplicador e as vezes ocorre a sedimentação gerando a separação do liquido e pigmento . ocorrendo assim falhas na aplicação.
`Poderiam me indicar um aditivo para auxiliar neste processo de eliminação desta separação.
=====================
Good morning, I would like to know if this same principle applies to a dispersion of pigments used in safety paints where the effect of the pigment is perceived through exposure to UV light and our big problem is the stabilization of sedimentation because the product cannot have a very high viscosity because in the application process, which is done through pens in very thin tubes and which lead the ink to the applicator nozzle and sometimes sedimentation occurs, generating the separation of liquid and pigment. thus failing the application.
`You could indicate an additive to assist in this process of eliminating this separation.