
Functional Pigments (FP) is a somewhat ambiguous term in the coatings industry because it can be interpreted to include anti-corrosive, conductive and special-effect pigments, as well as extenders (also known as inert pigments). This article focuses on these four types, but there are others.
Corrosion Inhibitive Pigments (CIP) help prevent or reduce the annual cost of steel corrosion, estimated to be over $400 billion in the United States and $2 trillion globally. Corrosion is primarily from electrochemical deterioration of a metal, due to the reaction with its environment, that transforms the metal into its lowest energy state. Oxidation occurs at the anode (positive electrode) and reduction occurs at the cathode (negative electrode). Corrosion is normally accelerated by the presence of water, oxygen and salts (particularly of strong acids).
These pigments have a minimum solubility, but to be active and effective, the coating must permit the diffusion of some water to dissolve the pigment. If the solubility is too high, the pigment will leach out of the coating too rapidly and reduce the time that the pigment is available to inhibit corrosion.
Another prime consideration in the selection of a corrosion inhibitive pigment is the pH. For example, a pigment with a high pH may have a deleterious effect on the cure of acid-catalyzed systems. Conversely, a pigment with a low pH may adversely affect the stability of waterborne systems.
The vast majority of corrosion inhibitive pigments are comprised of the combination of metal ions (cations) derived from zinc, strontium, chromium, lead, molybdenum, aluminum, calcium or barium and anions, such as those derived from phosphorous (orthophosphoric and polyphosphoric acids), chromic acid and boric acid. Although hexavalent chromate (Cr+6) and strontium are very effective in inhibiting corrosion, their use is very limited due to a variety of environmental and toxicological regulations.
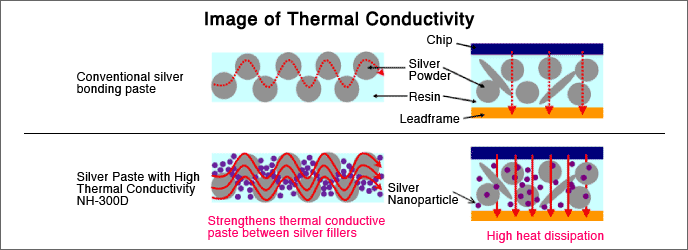
Conductive pigments (CP) are pigments that enable energy to be transported through a coating. Electrically conductive pigments allow electricity to be passed through or dissipated, as in static-dissipative materials. Thermally conductive pigments increase the rate at which heat transfers through a coating and have utilization in cooling systems, where an increase in heat loss through air conditioning fins and coils greatly reduces the energy costs to run the equipment. The types of pigments included in both applications include silver, nickel, silver-coated nickel, carbon black, multi-walled and single-walled carbon nanotubes, as well as other materials.
Some of these can be used to formulate coatings with EMI shielding. Electromagnetic shielding is the practice of reducing the electromagnetic field in a space by blocking the field with barriers made of conductive or magnetic materials. EMI-shielding is used with sensitive analytical instrumentation as well as protection of detection by radar.
Special-Effect Pigments (SEP) are pigments that provide an infinite array of colors and effects that enable unlimited design possibilities for coatings. These effects include the illusion of flickering lights, metallic reflection, interference sparkle and color variation and luster that changes with the viewing angle and light source. In a recent article in UL Prospector, this topic is thoroughly presented by Ron Lewarchik. The pigments reviewed are metallic flakes and thermochromic, photochromic, luminescent and phosphorescent pigments.
Extenders don’t sound very glitzy or important, but they are. They are used to provide many functions, including improvement in hiding, stain blocking, single-coat priming and painting and increased scrub resistance, to name a few. The use of small-particle silicates spaces the larger particles of titanium dioxide (TiO2) to improve the efficiency of the hiding pigment. Micaceous pigments are used in printing to provide better clarity of inks, as they won’t penetrate the paper substrate as much. In corrosion-inhibitive paints, micaceous iron oxide creates a “tortuous path” for water to migrate through the film, imparting improved protection.
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



Mark, nice article and visuals, you covered a lot of turf in a short article!
Ron, you made it a lot easier from your past articles – thanks!
Indeed a very informative article!
Can you please help pigments which can reflect in Far Infrared spectrum and can be dispersed in a highly alkaline system.
You would be best served if you were to contact a local producer of materials such as Ferro, Shepherd or Shanhai