Coatings consist primarily of binder (resin), as well as primary and extender pigments. They may also contain solvent, whether it’s water, organic or inorganic in nature. To a much lesser extent but sometimes more important, are additives.
Additives are typically used at levels of less than 5% in a formulation, but often have a significant impact on performance. An otherwise well-formulated coating can be ruined with the improper use of additives. In the additive world, “more is better” is almost never the case. Over the past decade, many new additives have been developed due to environmental regulations, utilization of green technologies and to keep pace with the new chemistries of binders.
Looking for coatings additives for your formulation?
Prospector has thousands of defoamers, adhesion promoters and UV protection materials!
Search Additives
It is not possible to review every additive in a brief overview, as they are too numerous. There are excellent articles with much detailed information on this topic. In a previous post on the Prospector Knowledge Center, surfactants were discussed. All surfactants are additives but not all additives are surfactants. Some additives do, in fact, affect surface properties, but others act within the body of the coating or are processing or application aids.
Table 1 provides an overview of many of the common additives, but it is not complete. Some of the ones listed can be included in more than one category, and readers may disagree with where some are included.
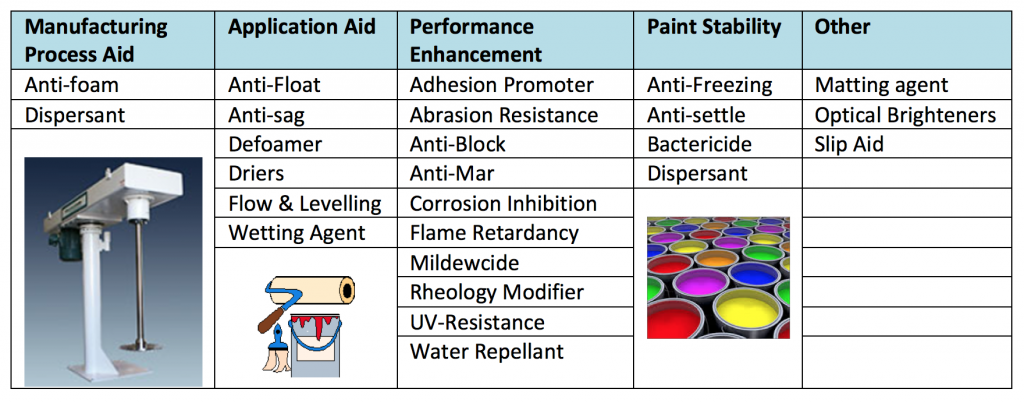
The three additive categories that will be presented are: defoamers/anti-foam agents, UV protection and adhesion promoters. These were chosen since their chemistries, applications and mode of activity are very diverse, and they can be some of the more challenging additives to employ in coatings formulations.
There is a distinction between anti-foam and defoamer. In the former, the intent is to impart a material that prevents the formation of entrapped air in the liquid coating during processing. This is typically most difficult in waterborne coatings since they possess relatively high surface energies at the liquid/air interface and do not release air easily. Anti-foams often perform best if they reduce the energies between small bubbles, so they agglomerate and when large enough, rise to the coating surface and break. Prevention of the bubble formation is, however, the most efficient activity.
A defoamer is a type of surfactant that breaks bubbles. Whereas anti-foams typically refer to materials used during processing, defoamers more often refer to their activity during application processes. Bubbles can be created by almost any application methodology, and particularly with air-assisted spray by roller. If the bubbles do not break quickly, there can be resultant film defects such as craters or pinholes.
There are many different types of defoamers that match the chemistries (waterborne vs. solvent or solventless) as well as application and finish. A formulator should be aware that a laboratory test to screen defoamers may not be relevant to an application. For example, if a wood coating with a strong defoamer is applied to a Leneta chart by brush, there may be instances of crawling or fisheyes. However, if the same coating is applied by spray, it may show no indication of film defects. The shear rate as applied by spray is far greater than as applied by brush, resulting in the disparity of film appearance. Rather than adding a surfactant to improve the appearance of the coating as applied to a card, if the formulator utilizes the correct application methodology, he can avoid the additional cost and effect of the addition of a “corrective” ingredient.
Rather than indicate UV protection by the traditional label of “UV-absorber/Hindered amine light stabilizer” or UV-A/HALS, the broader term of “protection” is used. Although the organic chemicals are most widely used for the protection from UV in sunlight, alternative inorganic chemistries are utilized in many applications, with the advent of nanomaterials. Particularly in exterior transparent or semi-transparent stains, nano-sized iron oxide, zinc oxide, cerium oxide and titanium dioxide, can be effectively employed without appreciably affecting clarity, color or gloss. In addition, the inorganic materials are far more permanent than the sacrificial nature of HALS/UVA. Nanomaterials are effective for UV-A, while other means are required for UV-B. The issues are similar for sunscreens for humans as well.
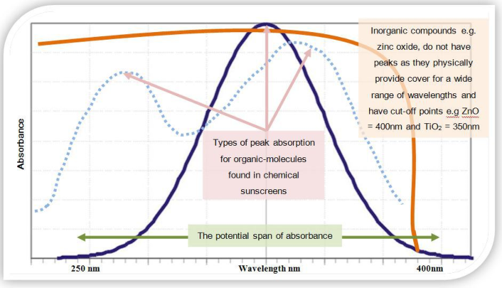
The principal ingredients in UV absorbers, are usually aromatic molecules conjugated with carbonyl groups. This general structure allows the molecule to absorb high-energy ultraviolet rays and release the energy as lower-energy rays (heat). Aromatic rings are degrade over time by radiation, and thus the organic absorbers are sacrificial in nature. Hindered Amine Light Stabilizers (HALS) are radical scavengers. Free radicals are released upon degradation of inorganic materials such as TiO2 and ZnO and these radicals degrade the polymer. HALS bind the free radicals and eliminate their effect. They are also sacrificial, similar to UV absorbers.
Adhesion promoters (AP) are materials that increase adhesive strength between the coating and the substrate, and sometimes are referred to as “coupling agents”. There are different chemistries that match the substrate as well as the solvent system. In addition, within a class of materials, the functionality on the backbone of the molecule will vary based on the resin system employed as well as the substrate to which it is attached. Silanes are effective AP’s for glass and silica-containing materials. Hence, they are used widely for Ultrahydrophobic treatments that rely on a system package of a silanol and silica, with silane adhesion used in epoxy, urethane and acrylic systems. Other adhesion promoters include titanates, chromiums, zircoaluminates and some monomeric phosphate chemistries. For plastic adhesion, some chlorinated polyolefin chemistries are effective in the improvement in adhesion of coatings to thermoplastic olefin (TPO).
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



I’m not sure what you are looking for. If this is a white coating, I suggest you use an exterior grade TIO2 to reduce chalking. If it is a clear coating, although it is a waterproof coating, you don’t specify if it is a water-based coating, or solvent-based coating. The recommendation is going to be based on the chemistry of the resin system, and water-based or solvent-based.