Heightened awareness of the importance of cleaning has led to an emphasis on automated systems for the decontamination of re-usable medical devices.

WASHtaK is a new product designed to manage, monitor and optimise clinical wash processes in hospital environments. Based on so-called tAK technology, it aims to ensure that post-operative infections from “unclean” instruments become a thing of the past. Currently, after a procedure, operators check for residues of blood on the instruments, but there has been no quantifiable way of ensuring the instruments are indeed “clean.”
The research which led to the creation of tAK technology began over 10 years ago at the Health Protection Agency (HPA) in the UK. At the time there was a significant concern about the emergence of variant Creutzfeldt–Jakob disease (vCJD) and its potential for spread through contaminated surgical instruments. vCJD can be transmitted via proteinaceous infectious particles, better known as prions. These prions adhere very well to stainless steel and are therefore very hard to remove in washing processes.
Scientists working at the HPA experimented with an enzyme known as thermostable Adenylate Kinase (abbreviated to tAK), which is isolated from organisms which can be found naturally around the very hot volcanic springs at the bottom of the ocean. tAK is a very robust and stable enzyme and was found to behave very much like prions do when coated onto a surface – very difficult to remove from the surface. The research team then developed this technology into a form that could be used to test the washing efficacy of washer disinfectors in hospitals’ sterile services departments. The basic idea is that, if one could measure the degree of removal of tAK from a test strip, then it would be possible to measure the washing efficacy of an automated washer disinfector (AWD) during routine use.

Accordingly, WASHtAK from Biotak features a tAK enzyme strip, sitting within a wash chamber. This is then analysed after use to assess whether the wash was indeed successfully “clean.” The core principle is that there is a known starting load of enzyme on the test strip, and the better the wash the less enzyme will be present afterwards. Contamination from handling the test strip can alter results, so in order to make this a viable concept, it needs an appropriate holder.
Rather than relying on objective comparison to a bottle label, as many such strip tests do, Biotak wanted to launch a fully traceable system, with quantifiable and immediate results. After considerable investment, the tAK analyser with a barcode scanner provides full traceability and a USB output for storing data.
Using the easy dispensing method designed into the WASHtAK holder, the uncontaminated strip can be taken straight from the wash, then scanned, assessed and provide a reading within minutes. This makes it possible almost instantly to know whether the wash is successful and if the instruments are indeed safe to use with patients.
“The whole aim is to give accurate, quantifiable results, straight after the wash, driving up standards and driving down costs for the NHS and other healthcare operators,” said Joanne Harmer of Biotak.
Malvern Healthcare was provided with a rough sketch of an idea for a hinged component that would house the strip safely in the wash and dispense it appropriately when required.
Designer Graham Robinson came up with a different component design that would make tooling more economical and provide all the holding and clipping features for each environment that it was being designed for. This eliminated over-complicating the tooling and component and produced a cost effective design for both.
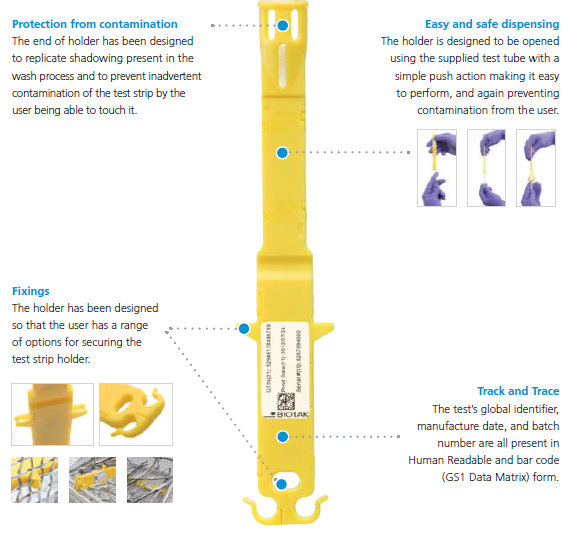
The end result was to make two halves of an injection moulding, with integral clipping features and a place for the enzyme coated strip to sit safely, whilst still sitting freely in the wash, but without risk of contamination by being touched.
After a prototype tool was manufactured, the product was tested by the Health Protection Agency by assembling small batch runs. Production was then moved to Andel Plastics when Robinson later became Technical Director there in October 2012. “I was really pleased that Biotak followed me in my new venture, as the product has always excited me and it’s so good to see a product go from concept, through all the testing and tweaking, into full production. It’s a really simple clever device that will solve a complex issue.”
Since then things have continued to develop with a production tool being manufactured at Andel Plastics’ in-house toolroom. The first major production run of this product has just been completed in 2Q 2015, ready for trials with the NHS and European Hospitals.
For the assembly of the enzyme strip into the new WASHtAK holder a Class 8 Clean room was required. The installation of the cleanroom took only three months from start to full certification and is now catering for two Medical Device customers.
In further developments, larger versions for chamber decontamination are planned, now the technology is proven and the quality assurance of the brand is there. Conceptually, the decontamination community has established the so called “SAL” – Sterility Assurance Level; in essence the SAL says a steriliser should be able to achieve sterility at the halfway point in its cycle, the idea being that this should give a pretty good safety margin. With WASHtAK, there’s no reason why health industry cannot start to work towards an equivalent WAL Washing Assurance Level.
ISO 15883 is the standard that is relevant to washer disinfectors. In addition to the quarterly and annual washing testing and validation, the current standard recommends routine testing of cleaning efficacy. It recommends daily tests to be carried out on the load (as opposed to just the chamber). It recommends using a simple soil test, but these soil tests have their shortcomings, namely that they are subjective and often very easy to clean meaning they don’t represent a realistic challenge.
References:
WASHtAK Demonstration
The Journal of Hospital Infection
Search for materials on Prospector…
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



Leave a Reply or Comment