
In April 2015, Prospector published an article on green resin technology using bio-based resin building blocks in the synthesis of polymers for coatings. Another highly desirable approach to green technology is the incorporation of resins utilizing building blocks derived from recyclable materials.
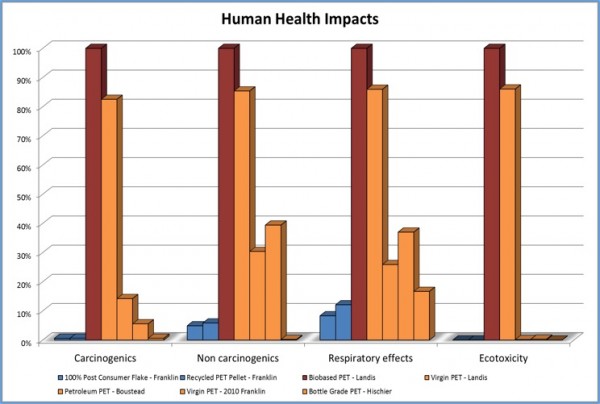
Sales for resins/polyols using recycled or bio-based polyols are expected to grow twice as fast as the overall polyol market in the next four years[1]. Driving forces for the use of recycled materials in the manufacture of resins can be reduced health hazard[2] (figure 2), as well as environmental and economic factors. Other attributes include reducing the carbon footprint[3] (Figure 1), increasing sustainability, and conservation of natural resources. Green products are also growing in favor with multiple government and private agencies.
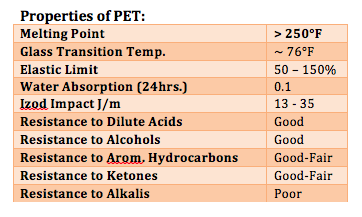
Typical recyclable material sources may include polyethylene terephthalate (PET), designated rPET for recycled PET, recovered cooking oils and recycled polyurethane foam. PET is typically used as containers for soft drinks and water, whereas polyurethane foams are used as carpet underlay and in mattresses. In the U.S. alone, there were 6.5 billion pounds of unrecycled PET-based containers in 2013.
The U.S. Environmental Protection Agency Comprehensive Procurement Guideline Program (CPG) defines recycled material as such that the EPA deems equivalent to virgin material. RCRA Section 6002 also requires purchasing agencies to establish procurement programs for designated items that meet CPG. Scientific Certification Services (SCS) recognizes products made either in whole or in part from recycled waste material in place of virgin materials. Through its certification process, SCS helps products qualify for credits within the LEED rating system. LEED is a certified U.S. Green Building Council program. Recycled content is certified by the U.S. Green Building Council’s GreenCircle for total recycled content based on pre and post consumer recycled content in products.

Various chemical processes are used to recover PET and polyurethanes including glycolysis [4], aminolysis, acidolysis and hydrolysis. For example, catalyzed glycolysis combines the recovered PET material[5] with diols and heat to produce a polyol for use in the synthesis of resins for use in coatings or other applications:

Several companies have developed polyols and resins from recycled materials for use in coatings, including Nippon Paint (polyester resins from rPET and recycled cooking oil), Emery and InfiGreen (polyols for coatings), and Terrin (polyols). Resinate Materials Group utilizes rPET to synthesize a multitude of polyols and resins using a number of different rPET based platforms with a recycled/renewable content as high as 99.9%, Resinate Materials Group technologies based on rPET include:
- Polyester polyols
- Polycarbonate polyols
- Polyols for 2K urethanes
- UV cure acrylates
- Ambient dry and UV Cureable PUD’s
- Plasticizers
A resin platform utilizing a high level of recyclable and renewable materials can offer not only the advantages mentioned previously, but can also provide enhanced performance. A polyol resin platform using rPET provides hydrolytic stability, solvent and chemical resistance, hardness, adhesion, flexibility and barrier protection to the migration of air and moisture vapor. An example of the physical performance that can be achieved in a coil coating formulation that utilizes a resin containing over 50% rPET is as follows:
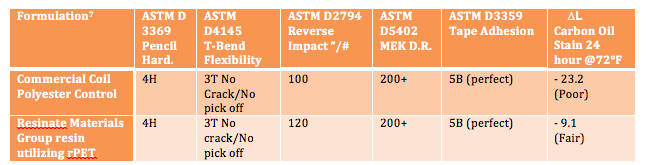
The use of recycled raw materials in resins and coatings is a growing area of interest as many companies are adding such materials to their product portfolio since there can be environmental, performance and economic benefits. Properly formulated coatings using recycled materials in their value stream can provide equivalent performance to their non bio-based counterparts.
Search for bio-based resins now…
[1] Resinate Materials Group overview slides, June 2015
[2] June, 2015 Coatings Technology, Resinate Materials Group
[3] June, 2015 Coatings Technology, Resinate Materials Group
[4] Scholars Research Library, Archives of Applied Science Research, 2012, 4 (1):85-93
[5] Polymers 2013, 5, 1258-1271; doi:10.3390/polym5041258
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



Leave a Reply or Comment