Avobenzone is the most important UVA absorber that has been globally approved. Avobenzone (EU) (formally 4-tert-butyl-4′-methoxydibenzoylmethane, or butyl methoxydibenzoylmethane) was patented in 1973 and commercialized by Hoffman LaRoche as Parsol 1789.
It was approved as a monographed sunscreen in the EU (up to 5%) in 1978 and by the FDA (up to 3%) in 1996. It is an oil-soluble, crystalline UVA sunscreen (EU) with a melting point of 81-86 C. Avobenzone has an absorption maximum of 357 nm and a UV absorption value (1% / 1 cm in methanol (EU) at 358 nm) of 1100 – 1160.
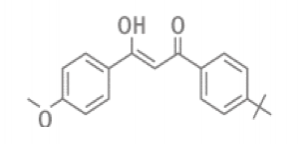
Avobenzone is a dibenzoylmethane derivative for which the ground state is a mixture of the enol/ keto forms. Only the enol form is an effective UVA absorber. When irradiated with UVA light, it generates a triplet excited state in the keto form that can cause the avobenzone to degrade, as well as cause deep-level skin damage. Avobenzone can lose up to 36% of its potential for UV absorption after only 1 hour of exposure to sunlight, if not stabilized.
Stabilizing Avobenzone
Perhaps the best approach to stabilizing avobenzone is to incorporate octocrylene (EU) in the formulation to help prevent the formation of the triplet state. Octocrylene is also a globally approved sunscreen (although some evidence suggests octocrylene may itself be a photo-sensitizer, causing indirect DNA damage). The key L’Oreal patents (in the United States) for stabilization of avobenzone using octocrylene expired in late 2014 and are now freely available.
This approach can help you formulate avobenzone with octinoxate, which can react with avobenzone via a 2+2 cycloaddition reaction to form a non-absorbing chemical. Titanium (EU) and zinc oxide (EU) can have a negative effect on avobenzone stability unless properly coated. However, these combinations aren’t permitted in the U.S. under the current U.S. monograph.
Metals such as iron (EU), aluminum (EU), or zinc (EU) can cause discoloration, loss of UV absorption, and reduced solubility if not complexed. Chelating agents should also be used to prevent this type of interaction with multivalent metals. Formaldehyde (EU) donating preservatives should also be avoided.
Recommended Photo Stabilizers:
- Octocrylene (BASF)
- Octocrylene (DSM)
- Corapan® TQ (Diethylhexyl 2,6-Naphthalate; Symrise)
- Oxynex® ST ( Diethylhexyl Syringylidene Malonate; EMD)
- Synoxyl® HSS (Trimethoxybenzylidene Pentanedione; Sytheon)
- SolaStay® P1 (Polyester-25; Hallstar)
- Polycrylene® (Polyester-8; Hallstar)
- SolaStay S1 (Ethylhexyl Methoxycrylene; Hallstar)
(Products available in Europe: Octocrylene (BASF) | Octocrylene (DSM) | Corapan® TQ (Symrise) | Synoxyl® HSS (Syntheon) | SolaStay® P1 | Polycrylene®, SolaStay® S1 (Hallstar))
Avobenzone, as a crystalline material, must be completely solubilized in order to be effective. Liquid sunscreens like homosalate (EU) (avobenzone – 19% soluble) and octyl salicylate (EU) (avobenzone – 21% soluble) are poor UVB sunscreens, but are good solvents for avobenzone and oxybenzone. Octinoxate (octyl methoxycinnamate) can dissolve up to 25% avobenzone.
A simple way to quickly screen formulations is to make only the liquid oil phase of the formulation and test the samples at low temperatures for crystallization.
Examples of non-sunscreen solvents (approximate solubility of avobenzone in solvent):
- 40%-Butylphthalimide Isopropylphthalimide (Pelemol® PIP; Phoenix Chemical)
- 32%-Ethylhexyl Methoxycrylene (Solastay® S1; Hallstar)
- 24%-Phenyl Ethyl Benzoate (X-Tend™ 226; Ashland)
- 20%-Dioctyl Isosorbide (Synovea® DOI; Sytheon)
- 20% -Isopropyl Lauryl Sarcosinate (Eldew® SL 205; Ajinomoto)
- 18%-Ethyhexyl Benzoate (Finsolv® EB; Innospec)
- 18%-Phenoxycaprylate (Tegosoft® XC; Evonik)
- 16%-C12/15 Alcohol Benzoate (Finsolv TN®; InnoSpec)
(Products available in Europe: Phoenix Chemical | Solastay® S1 | X-Tend™ 226 (Ashland) | Synovea® DOI | Eldew® SL 205 (Ajinomoto) | Finsolv® EB (Innospec) | Tegosoft® XC (Evonik) | Finsolv® TN)
Material Resources in Prospector:
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



George,
Is it allowed in the US to use pigmentary Titanium Dioxide to color a product such as a lipstick that contains Avobenzone as an active sunscreen ingredient?
Hi George
Is it allowed in the US to use pigmentary Titanium Dioxide to color a product like a lipstick or foundation that contains Avobenzone as an active sunscreen ingredient?
Good day, Charlie.
To answer your question, no, it is not allowed.
Thank you for reading!
George