A critical part of any coatings formulation is ensuring that the coating will be free of inherent defects, including pigment flooding and floating. Waterborne formulations represent some unique challenges due to multiple factors, including the high surface tension and polarity of water that does not contribute to the wetting of most pigment and filler particles. In this article, I’ll define some important considerations in formulating waterborne paints to avoid pigment flooding and floating.
Flooding, Floating, Surface Tension, Bernard Cells, Flocculation and Agglomeration
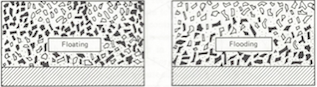
Floating describes a mottled, splotchy appearance on the surface of a paint film. It is most apparent in coatings colored with two or more pigments and is a result of the horizontal separation of different pigments. Flooding is the phenomena observed when the surface color of an applied film is uniform but is darker or lighter than it should be. This is attributed to a vertical separation of different pigments in the film.
Surface Tension results when the force that occurs in a liquid at the interface differs from the forces within the liquid. Thus, surface tension is caused from the surface molecules having a higher free energy than those molecules in the bulk of the liquid.
Surface tension differential can cause a convection current resulting in a regular hexagonal surface pattern called Benard Cells. A hexagonal Benard Cell pattern results in smaller, more mobile pigment particles (smaller, less dense) being deposited on the perimeter and the less mobile particles (larger, more dense) remaining away from the perimeter.

Flocculation is the recombination of dispersed pigment particles that were not properly stabilized in the pigment dispersion. Flocculation is undesirable at it detracts from hiding and color development. Flocculation is reversible by applying a low degree of shear. In figure 2, the phthalocyanine (EU) blue pigment is flocculated. Upon rubbing with a finger, the deeper blue color returns.
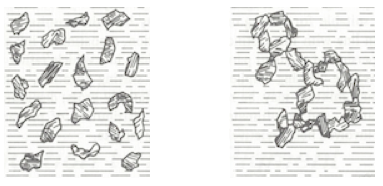
Pigment agglomeration is defined as pigment particles that are clumped together without sufficient vehicle or wetting agents present between pigment particles. When agglomeration occurs, extensive shear and attrition forces are usually necessary to reinstate a stabilized pigment dispersion.
Remedial Actions to Overcome Floating and Flooding in Waterborne Paints
Pigment dispersion in aqueous media uses the same principles as in organic solvent media such as proper wetting, pigment dispersion and stabilization. However, the surface tension of water and high polarity make it more problematic in wetting low polarity pigments. In many cases, water interacts aggressively with the surface of the pigment, destabilizing the dispersant on the pigment surface. Overcoming flooding and floating starts with selecting pigments that are free of fines. Also, many pigment manufacturers supply surface treated pigments to avoid flooding and floating.
Secondly, ensure that the pigment dispersion is uniform and stabilized (elimination of pigment flocculation of one pigment, with the exclusion of other pigments).
Thirdly, the use of suitable wetting agents/surfactants help to ameliorate differences in polarity and surface tension between pigments that contribute to flooding and floating. Inorganic pigments such as iron oxides (EU), titanium dioxide (EU), calcium carbonate (EU) and many other filler pigments have a polar surface and are easily wet by water. However, water alone normally does not stabilize the pigment dispersion against flocculation, so they require a surfactant to wet and stabilize the dispersion. Also, many pigment manufacturers supply surface treated pigments to avoid flooding and floating. For some organic pigments that have a surface with low polarity, many manufacturers modify the surface with a layer of inorganic oxide to provide increased polarity to improve wetting in aqueous-based systems.
Fourthly, the use of an appropriate thixotrope (EU) helps to build sufficient viscosity and a network structure that discourages pigment separation. However, one must be sure that there is acceptable compatibility between the thixatrope and dispersant (EU).

Overcoming flooding and floating in solvent-borne paints primarily involves utilizing a suitable pigment dispersant and the elimination of Benard Cell formation with the addition of a surface control agent.
A final consideration affecting the stability of an aqueous paint pigment is the pH of the pigments. For example, if a low pH carbon black pigment is used in the pigment dispersion without the use of a suitable wetting agent in an anodic aqueous based paint (pH normally > 8), longer term instability can result as the neutralizing amine on the resin backbone can migrate to the acidic pigment.

Care must be taken while reducing a latex paint with water because floating can occur (slow addition of water with proper mixing and ensure against over reduction). This is due to a shift in the equilibrium between the dispersed pigment particles and water, resulting in a decrease in the amount of stabilizing dispersant on the pigment.
There are numerous suppliers listed in UL’s Prospector website with a wide variety of wetting agents (EU), pigment dispersants (EU), surfactants (EU), thixotropes (EU) and pigments to meet your requirements in both solvent and waterborne coatings.
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



I am experiencing flocculation on my water-based latex paint formulation. What do i need to add to prevent this problem?
Hello Alex,
Please provide additional information in terms of the paint color, the pigments and vehicle type you are using. Secondly, at what step of the paint making process do you observe the flocculation?
I don’t know if you can help me. I am looking for the correct combination of vehicles to allow a silica based pigment to float on water. The density of the pigment solution and the viscosity of the water has me stumped. I’ve tried lots of things. I truly appreciate anything you can tell me. Thank you so much.
Hello Barbara,
Silica vary in particle size, particle size distribution, surface area and surface treatment. As I do not know what specific silica you are using I suggest that you contact the supplier to find additional specifics as well as a recommendation. In general a smaller particle size silica with a large surface area and appropriate surface treatment (e.g. wax) or other lypophilic surface treatment will offer the best chance to “float” the silica. Proper d
ispersant selection to provide optimum compatibility with the silica surface treatment will also enhance your chances of observing a positive result.
Floating is vertical separation as we can see everywhere and on your diagram but you say :” floating is a result of the horizontal separation of different pigments” and i agree with this sentense.
Flooding is horizontal séparation but you say :”This is attributed to a vertical separation of different pigments in the film”
so i don’t undersand
Hi there I would like to make the best tattoo in can you help me with that ?
I am experiencing FLOTATION issues in our Yellow Oxide dispersion used for WAter-based emulsion paints. What do we need to add to prevent it?
I am interested to make and epoxy and a polyurethane super hydrophobic. Can you advise on suitability of Silicon Oxide (SiO2) Nanopowder, Coated with 2wt% Silane for either one?
Providing a superhydrophobic surface involves the proper placement of the agent that provides superhydrophobicity, the selection of the material providing superhydrophobicity as well as the formulation that it is used in. Feel free to contact us directly for consulting services as the answer to your question has multiple layers and considerations.
Hellow sir! Today I got a problem after dispersing the hans scarlet orange pigment in a product called Hi chemical enamel orange whereby epoxy used as resin and xylene used for high amount compared as butly acetate and butly glycol also neusperse was used as surfactant while bentone used as wetting agent. The problem comes the paint/product become separate each other what there is no uniform as a product, what could be the reason? Please help me! Am a young paint chemist fresh from University in Tanzania!
Dear Said:
It looks like the resin in your paint is not compatible in the solvent blend you are using, hence you are seeing a phase separation. Is the HI chemical enamel you are using an alkyd paint? If so depending on the ratio of solvents in your formulation, solvent compatibility with alcohol solvents and the alkyd paint may be the issue.
Thank you for reading!
Ron
We prepared and coated a 500 M2 warehouse floor, with a blue water based epoxy, it fully cured and the adhesion is good. However now some15 months after application, the surface has been walked over and wheeled trucks have passed over the surface.
Now there seems to be a blue dust rising from the coating, onto shoes and wheels. We have never seen this before, is it possible that it could be floatation of the blue pigment in the epoxy. if not any idea’s
Please complete the contact form on the ChemicalDynamics.net web site and we’ll get a response to you soon.
Thank you for reading,
Ron