Dry versus conditioned nylon explained
Standard nylon materials are hygroscopic (absorb moisture from the air) and are capable of absorbing upward of 8% moisture by weight at saturation (compared to 0.8% for acetal).
This is a completely reversible physical reaction. The higher the humidity, the faster nylon will absorb moisture. Immersing in water will cause it to absorb moisture extremely fast. However, it only absorbs moisture until it is saturated. At that point, no amount of humidity will make it absorb more. Like a sponge, once the sponge has been soaked in water to saturation, it will not hold more water. Also like a sponge, when nylon is exposed to dryer air, it will give that moisture up and dry out.
Under normal conditions, say 20-80% RH, nylon will reach saturation in about 24 hours. This means that the same amount of drying time will be required to dry material that has sat open for 24 hours or 24 years.

Dry and conditioned data
Nylons are crystalline polymers, but some amorphous regions do exist. It is from these crystalline regions, which resist being pulled apart, that polyamide receives much of its strength and stiffness.
But when water molecules contact the nylon structure, they diffuse though the material and force the polymer chains apart. This is the reason why polyamide parts swell after being exposed to moisture and they exhibit diminished mechanical properties. It is within the amorphous regions that the water bonds to the polymer chain. The crystalline regions of polyamide resist being pulled apart by the water because the bonds between the amide groups are stronger than the attraction to water. Were this not the case polyamide would dissolve in water. Instead, water acts as a plasticiser.
Plasticisers cause a polymer to swell and soften. Both effects are exhibited by polyamides when they are exposed to moisture and must be considered when designing a part. Hence why both dry and conditioned data for polyamides are given.
What is the difference between data quoted for dry and conditioned data for plastic materials? And why is this most significant for nylons (polyamides or PA)?
Dry: Data that is obtained from a sample of material with equivalent moisture content as when it was moulded (typically <0.2%).
Conditioned: data obtained from a sample of material that has absorbed some environmental moisture at 50% relative humidity prior to testing.
Effect of moisture on properties
Moisture acts as a plasticizer in nylon and therefore reduces strength and stiffness properties but increases elongation and toughness. The mechanical properties of nylon are likely to change over time, due to moisture uptake. In general, as moisture content rises, significant increases occur in the impact strength and other energy absorbing characteristics of the material. The glass transition temperature reduces, dropping by 20°C or more.
Conversely, if nylons are used within a dehumidified atmosphere, the material can dry out, leading to embrittlement and cracking.
Addition of fillers reduces the effect of moisture. Reinforcements reduce the effects more than fillers.
If a part is exposed to intermittent periods of moist and dry environments, it will continue to absorb and give up moisture and the properties will change accordingly for the life of the part. It will never reach any type of equilibrium. Drying in an oven will drive off all but a small percentage of the water molecules, while a part that is outside on a hot sunny day could certainly release a large amount of its moisture.
Dimensional stability
The dimensions of nylon parts are dramatically affected by temperature and humidity. This can severely affect the functionality of long parts. It is important to take these issues into account when designing a nylon part. If the change in dimensions is unacceptable, it is prudent to look instead at acetal or thermoplastic polyesters (PBT or PET) as alternative materials.
At room temperature in a 50% relative humidity environment, equilibrium moisture content for nylon tends to remain around 2%, which corresponds to an increase in size of roughly 0.5 – 0.6%. Under similar conditions, acetal is expected to absorb roughly 0.2% moisture by weight and grow around 0.2%.
Variations between nylon grades
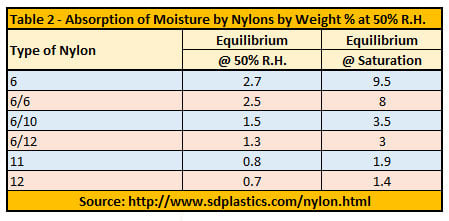
The extent to which the different properties change depends a great deal on the chemistry of the polymer itself. There are many nylon variants*: these include nylon 6, 6/6, 6/9, 6/10, 6/12, 4/6, 11 and 12, semi-aromatic polyarylamides (PARAs) and polyphthalamide (PPA high performance polyamide). Polyamide 12, for example, doesn’t absorb as much moisture as Polyamide 6, so Polyamide 12’s properties don’t fluctuate as much with moisture.
Table 2 shows how the absorption of moisture varies with different nylon grades. The characteristics of these various grades is covered in more detail in this article.
Processing
Moisture in the raw material causes many processing issues and part failures, while the properties of nylon mouldings are directly affected by the amount of moisture they contain.
Nylon drools and flashes if it is injection moulded wet. The water will reach boiling point at the processing temperature of the material. That liquid water expanding into steam is very destructive to the polymer, causing loss of properties and the splay appearance that we are all familiar with.
Drying before moulding
Moisture absorbed onto the surface of the nylon resin or part can generally be removed by drying for three or four hours. No amount of drying time or temperature will get that water to evaporate. However, for moisture that has absorbed more deeply, a much longer drying time is needed, sometimes up to 24 hours or more, to allow sufficient time for the water molecules to diffuse to the surface. Most nylon manufacturers will recommend drying nylon to below 0.2% moisture content. This will typically yield acceptable parts — reducing the moisture even further will improve the properties still more.
The only exception to this would be if the material were compounded in such a way that a large amount of water gets encapsulated in a small hollow spot in the pellets. This is called encapsulated moisture, though it is rare.
However, moisture in nylon also acts as a lubricant that increases the flow of the material. The higher the moisture content, the higher the flow. Melt flow is not reported on a nylon data sheet because the moisture content cannot be controlled well enough to provide meaningful data.
If the material is dried beyond 0.2%, the flow decreases that can then make it difficult to fill the mould cavity. If moulders then alter their processing parameters to try and fill the cavity, they can end up degrading the material with shear heat.
* Common variants include Nylon 6, Nylon 6/6, Nylon 66, and Nylon 6/66. The numbers indicate the number of carbon atoms between acid and amine groups. Single digits, like “6”, indicate that the material is devised from a single monomer in combination with itself, i.e., the molecule as a whole is a homopolymer. Two digits, like “66”, indicate that the material is devised from multiple monomers in combination with each other (comonomers). The slash indicates that the material is made up of different comonomer groups in conjunction with each other, i.e., it is a copolymer.
UL Prospector’s Ben Howe contributed to the content for this article in its original publication.
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.


