By Mike Kayat, Intevac Photonics, DeltaNu
The global medical plastics industry is growing at an increasing annual volume. Industry drivers are principally the development of new medical devices that replace metal parts with advanced polymers, the increased use of these devices for an increasing number of longer living patients in both developed and emerging countries, and the move to disposable products. The increasing availability of portable, point of care devices is also a key development driving home-based diagnostic applications. About three-quarters of the medical plastics market is comprised of pharmaceutical and biological products, the rest of the market is made up of surgical appliances and medical instruments.
How large is the market for medical plastics? For the US, recent research reports estimate that by 2011, over 3 billion pounds of medical grade plastics will be utilized, growing to over 4 billion pounds in 2015. Globally, some 15 billion pounds will be consumed in 2015, as both developed and emerging countries use more medical devices. Commodity resins including PE, PP, PVC, PS and PET, make up over 50% of medical plastics used, engineering plastics including nylon, PC, ABS come in at about 15% with thermoplastic elastomers such as TPU, TPV and SBC making up the rest. A wide range of additives are typically used in compounds.
Stringent Quality Control Requirements
Patient safety drives the adoption of government regulations. Medical grade plastics must be long lasting, be able to be sterilized with radiation, exhibit blood compatibility, particularly antithrombotic activity and other characteristics which are symbiotic with the human environment. In the US, the FDA mandates close communications between manufacturers and suppliers. However, problems can be created when even slight changes are made in approved materials. Timely incoming inspection of raw materials, by manufacturers is a critical step in ensuring a rigorous QA/QC process, to avoid potential brand damage with high recall costs.
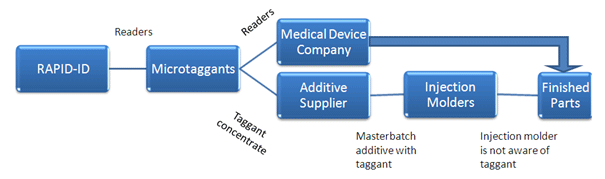
Now, there is an easy, cost-effective, palm-sized solution that significantly reduces the incidence of misidentification: RAPID•ID™, designed by DeltaNu (www.deltanu.com), the leader in handheld materials identification instruments. This tool has been used to support FDA mandated QA/QC processes within the medical plastics industry, along with other industries.

Figure 1: RAPID•ID™ materials identifier. |
The plastics market has numerous methods to assist in identifying incoming materials like the burn and sniff test and density tests. They’re time consuming, not always the most practical, and most end up destroying the sample. These tests also create many challenges when trying to quickly, and reliably identify plastics, but there are alternative methods available that are safer and faster. With several growing concerns in the plastics market, including phthalates in children’s toys, a quick dependable option is becoming a necessity. |
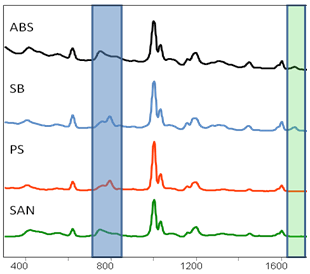
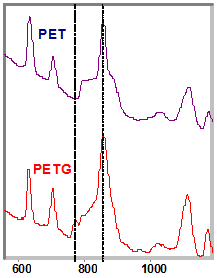 |
Figure 2: RAPID•ID™ pinpoints the minute optical differences in plastic materials, for example, resins and styrenics. |
Additives and Counterfeits
A number of additives are typically included in medical plastics products during the molding or extrusion processes. These include: anti-static agents, lubricants, plasticers and stabilizers. These may be contaminated or have variable concentrations, leading to adverse effects when used in end user environments. A quick, reliable approach is required to identify these additives and confirm or otherwise, their presence in target materials.
Another major issue facing manufacturers concerns counterfeit materials. Recent product recalls cite a range of counterfeit medical plastics components that were initially difficult to detect and identify. Once again, a quick, reliable solution is needed to isolate counterfeit parts and materials.
Authentification using Taggants
With a focus on enhanced security solutions to prevent counterfeit activities, along with more sensitive QA/QC control, several companies have developed microscopic chemically active materials now called taggants. These taggants can be uniquely encoded and added to a wide range of dry and wet product materials. Taggants can be used for a yes/no or pass/fail identification procedure, or for verifying that certain materials have not been substituted or diluted, or are missing altogether.
One company, Microtrace, LLC. (www.microtracesolutions.com), manufactures several different types of taggants ranging from microscopic particles called Microtaggant® to taggants which are completely soluble and invisible, which are called Molecular taggants. Microtrace supplies its taggants to the plastics industry in two delivery formats. For the compounder or masterbatcher, the Microtaggants are delivered in a dry particle form while the Molecular taggant is delivered as a masterbatch concentrate. For the plastic processor, both the Microtaggant and Molecular taggant are delivered in a finished masterbatch per specification, ready for processing typically at a standard 1% – 2% let down rate. Figure 3, shows a typical Microtaggant material that can be added to a typical resin masterbatch, shown in Figure 4.
Microtrace taggants enable finished products to contain covert authentification capabilities for product security and brand protection.
|
Figure 3: Magnified image of Microtaggants. |
Figure 4: Microtrace masterbatch with taggants. |
| A typical scenario for medical plastics may the following. A medical device company specifies an additive to be incorporated into molded parts by their contract manufacturers. The company needs the ability to quickly check parts in the field to determine that each manufacturer has (1) used the correct and specified additive and (2) that the additive was used at the correct loading rate in the final part, Figure 5. In addition to confirming the additive and loading rate, the medical device company can verify part authenticity, along with production level of each contract manufacturer to prevent unauthorized production or overruns. | |
|
Figure 5: RAPID•ID™ verifies the corrective additive, additive loading along with authenticity. |
|
| The Microtrace taggants are compatible with carriers like inks, paints and polymers and produce a strong optical signal that can be “read” by the RAPID•ID™ in a simple to use operation, Figure 6. Microtrace taggants can be incorporated into materials at extremely low concentrations making tagging of large volumes of bulk material very economical. | |
|
Figure 6: Simple to use operation to detect the presence of the Microtrace taggant. Microtrace can provide multiple taggant technologies in a single delivery format. This enables several levels of authentification, along with robust identification of medical plastics materials. RAPID•ID™, Prescription for Medical Plastics RAPID•ID™ has quickly become a valuable quality control tool for materials inspection when used together with DeltaNu’s advanced matching software. This precision system enables companies to better understand the reasons why materials that seem identical behave differently than expected and will give you the technology to ensure that your suppliers are in complete compliance with specified formulations. The advanced libraries are easy to set up and will work both on the laptop PC software and the unit itself in the field and can be modified to accommodate changes in the material specifications. RAPID•ID™ is a vital tool for making sure a medical device company received the exact material they order. Coupling the taggant technology with RAPID•ID™ enables highest levels of QA/QC and security to be implemented. For more information about how RAPID•ID™ can quickly and accurately medical plastic materials while supporting stringent FDA processes, please call 1-866-301-6328 or email sales@deltanu.com |
|
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.