 Steel can be protected against corrosion by using organic coatings1,2. Industries make the transfer from solvent-based systems to paints that are more friendly with respect to health and environment. This article is about water-based protective coatings that are used to protect steel against corrosion. To be more precise: below, I will discuss the general composition of a category of water-based anti-corrosion (AC) primers based on epoxy-amine chemistry.
Steel can be protected against corrosion by using organic coatings1,2. Industries make the transfer from solvent-based systems to paints that are more friendly with respect to health and environment. This article is about water-based protective coatings that are used to protect steel against corrosion. To be more precise: below, I will discuss the general composition of a category of water-based anti-corrosion (AC) primers based on epoxy-amine chemistry.
Corrosion of steel
Corrosion of steel (rusting) is an electrochemical process in which iron (Fe) in steel oxidizes into iron oxide (Fe2O3), also called rust. Both oxygen (O2) and water (H2O) must be present for rusting to take place. The undesired phenomenon is promoted by salts and acids. Corrosion protection of steel can be based on a few principles. First, and most important, the steel surface is passivated. This is done by assuring that a protective layer is formed on the surface of the steel. This so-called passivation layer prevents corrosion to continue. Secondly, the transport of molecules, that cause and catalyze corrosion, through the coating is minimized. It is said that the coating system forms a barrier for such molecules.
Cathodic protection, for example by using zinc particles as pigment, is a third concept that can be used to prevent rusting.
The coating system
A simple coating system for steel consists of 2 layers that are applied on steel that is properly pre-treated, Figure 1.

The function of the first layer, the primer, is to provide adhesion of the coating system on the steel and to protect the steel against corrosion. The second layer gives the desired aesthetical properties, like gloss and color, to the coated object. The topcoat also protects the object and the primer against external influences like UV radiation, mechanical punishment and rain.
We focus on the anti-corrosion (AC) primer.
Primer formulation
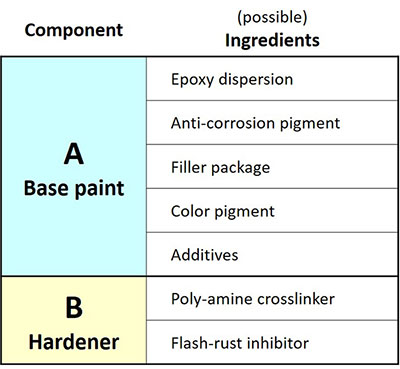
An example of the composition of a modern water-based, 2-component, AC-primer is given in Table 1.
The primer consists of 2 parts, the base paint and the hardener, that are mixed shortly before the paint is applied.
The binder system
Epoxies, paints based on epoxy-amine chemistry, is a dominant category of anti-corrosion primers. Key advantages of epoxy-amine systems are: excellent adhesion on metals, good mechanical properties and good resistance against water, solvents and chemicals. The AC-primer of Table 1 is based on a water-based dispersion of an epoxy-functional resin and a water-based dispersion of an amine-functional crosslinker, being the core ingredient of the hardener. After mixing, the dispersions coalesce and the epoxy and amine groups crosslink, thus forming a strong three-dimensional chemical network.
Anti-corrosion pigment
Effective AC-pigments form a passivating layer on the steel surface, thus stopping corrosion.
Hazardous anti-corrosion pigments, that are based on lead and hexavalent chromium (Cr VI), are more-and-more replaced by pigments that have a more favorable profile with respect to health and environment. Modern AC-pigments, mostly based on polyphosphate chemistry, are slightly soluble in water. The ions, released during dissolution, migrate to the steel-primer interface and form a protective phosphate layer on the steel surface. An example of such a pigment for water-based primers for steel is Heucophos® CAPP, a zinc-free pigment based on calcium-aluminium polyphosphate chemistry.
The primer does not contain a pigment that gives cathodic protection.
Fillers
Many steel primers contain a mix of fillers. Preferably, the fillers do not contain water-soluble salts.
A platelet-shaped filler is present in many AC-primers. The shape of these particles provides the so-called roof-tile effect, implying that the platelets overlap each other, thus providing improved barrier properties, Figure 2. An example of such a filler is Mica SG.
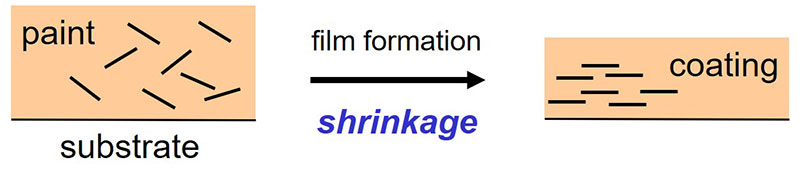
Some fillers crosslink together with the epoxy-amine binder system during film formation. In this way a stronger coating with improved barrier properties is obtained. Tremin 283-600 EST, for example, is a wollastonite filler that is surface-treated with an epoxy-functional silane. The silane parts of the molecules provide chemical bonds with the surface of the filler particles and the epoxy groups crosslink together with the amine groups of the crosslinker during film formation. In this way, the filler particles become part of the three-dimensional chemical network.
The rest of the filler package consists of inert inorganic particles, like barium sulphate.
Color pigment
For white primers, a high-quality titanium dioxide pigment with low water-soluble ion content, like Ti-Pure™ R-900, is preferably used.
Additives
Depending the exact formulation, several additives can be present in the base paint. A defoamer prevents the stabilization of air bubbles, a dispersant gives stability of the pigments and fillers against flocculation and a rheology additive prevents sedimentation during storage.
A coalescing agent may be needed to assure sufficient coalescence and merging of the epoxy and polyamine particles during film formation.
An adhesion promotor may be used to improve adhesion when, for example, smooth steel must be coated.
Flash-rust inhibitor
A problem that often occurs when water-based paint is applied on steel is that immediate corrosion takes place. This, so-called flash-rust, is prevented by using a flash-rust inhibitor as additive in the primer formulation. Raybo 80 NoRust is a water-based solution of high pH, based on sodium nitrite (NaNO2).
The future
A development in coating industry is to substitute the two coating layers (primer + topcoat) by only one coating. These so-called direct-to-metal (DTM) coatings provide all desired properties (adhesion, corrosion protection and protection against external influences) in one layer3.
Flash-rust inhibitors based on sodium nitrite have an unfavorable health and environment profile. Substituting products are under development.
References
- Article The Chemistry of Corrosion Protection and Anti-Corrosion Coatings, Wally Kesler, 29 September 2017, Prospector Knowledge Center.
- Article Fundamentals of Corrosion Protection, Ron Lewarchik, 2 October 2015, Prospector Knowledge Center.
- Article The Rapidly Growing Segment of Direct to Metal Coatings, Ron Lewarchik, 12 July 2019, Prospector Knowledge Center.
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



Love the way you provide the info in depth, Thank you for sharing this blog….
very informative article. I am more knowledgeable about primer because of this
thanks for sharing this! atleast now I know whats the use of primer and how it works
Thank you for posting this awesome blog post, really wonderful reading this.