Surfactants play an important role in many processes of interest in both fundamental and applied science, such as cleaning, wetting, dispersing, emulsifying, foaming and anti-foaming. One important property of surfactants is the formation of colloidal-sized clusters in solutions known as micelles. Surfactants are found in many products including paints, emulsions adhesives, inks, biocides (sanitizers), shampoos, toothpastes, firefighting (foams), detergents, insecticides, deinking of recycled papers, ski waxes, pharmaceutical applications in modifying existing medical preparations.
play an important role in many processes of interest in both fundamental and applied science, such as cleaning, wetting, dispersing, emulsifying, foaming and anti-foaming. One important property of surfactants is the formation of colloidal-sized clusters in solutions known as micelles. Surfactants are found in many products including paints, emulsions adhesives, inks, biocides (sanitizers), shampoos, toothpastes, firefighting (foams), detergents, insecticides, deinking of recycled papers, ski waxes, pharmaceutical applications in modifying existing medical preparations.
Surfactants are materials that lower the surface tension (or interfacial tension) between two liquids or between a liquid and a solid. In the general sense, any material that affects the interfacial surface tension can be considered a surfactant, but in the practical sense, surfactants may act as wetting agents, emulsifiers, foaming agents, and dispersants in coatings.
In the bulk aqueous phase, surfactants form masses, such as micelles, where the hydrophobic tails form the core and the hydrophilic heads are immersed in the surrounding liquid. Other types of structures can also be formed, such as spherical micelles or lipid bilayers. The shape of the molecules depends on the balance in size between hydrophilic head and hydrophobic tail. A measure of this is the HLB, Hydrophilic-lipophilic balance. Higher HLB surfactants (>10) are hydrophilic (“water loving”) and form O/W (oil-in-water) emulsions. Lipophilic surfactants possess low HLB values (1-10) and form W/O (water-in-oil) emulsions. Dish detergents, surfactants for emulsion polymerization, and SLS (Sodium Lauryl Sulfate) are high HLB surfactants.

The dynamics of surfactant adsorption are of great importance for practical applications such as in emulsifying or coating processes as well as foaming, where bubbles or drops are rapidly generated and need to be stabilized. As the interface is created, the adsorption is limited by the diffusion of the surfactant to the interface, which can result in the kinetics being limited. These energy barriers can be due to steric or electrostatic repulsions; steric repulsions form the basis of how dispersants function. Surface rheology of surfactant layers is important to the stability of foams and emulsions.
Most surfactants’ “tails” are fairly similar, consisting of a hydrocarbon chain, which can be branched, linear, or aromatic. Fluorosurfactants have fluorocarbon chains. Siloxane surfactants have siloxane chains. Recent advances in surfactant technology have seen the development of mixed chains or/and complex structures.

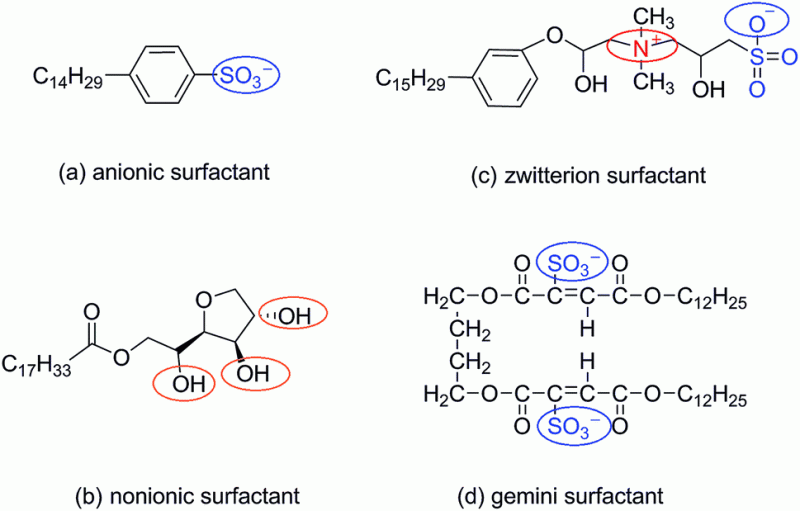
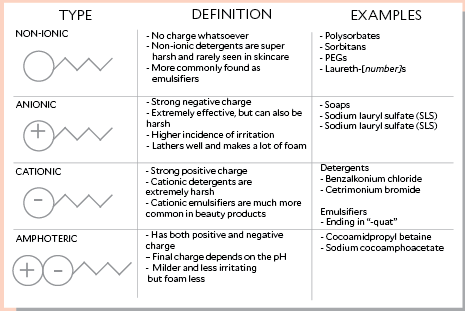
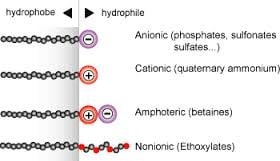
There are 4 types of surfactants (nominally) with a brief review of each as follows. These classifications are based upon the composition of the polarity of the head group: nonionic, anionic, cationic, amphoteric.
A non-ionic surfactant has no charge groups in its head. The head of an ionic surfactant carries a net charge. If the charge is negative, the surfactant is more specifically called anionic; if the charge is positive, it is called cationic. If a surfactant contains a head with two oppositely charged groups, it is termed zwitterionic. Commonly encountered surfactants of each type are listed as follows. A complete compendium can be found on www.ULProspector.com.
Nonionic surfactant
Nonionic surfactants are surfactants that contain ether [–(CH2CH2O)nOH] and/or hydroxyl [–OH] hydrophilic groups. They make up the largest group of surfactants. Unlike anionic and cationic surfactants, non-ionic surfactants are nonelectrolytes; that is, their hydrophilic groups do not ionize at any pH value.
Non-ionic surfactants are commonly used for stabilizing oil-in-water (o/w) and water-in-oil (w/o) emulsions. Since the non-ionic surfactants do not contain an ionizable group, their properties are much less sensitive to changes in the pH of the medium and the presence of electrolytes. In addition, they have fewer interactions with cell membranes compared with the anionic and cationic surfactants. Thus, non-ionic surfactants are preferred for oral and parenteral formulations because of their low tissue irritation and toxicity.
Examples of non-ionic surfactants include sorbitan fatty acid esters, (Spans®), polysorbates (Tweens®), and poloxamer (Pluronics®). Sorbitan fatty acid esters such as sorbitan monopalmitate are oil-soluble emulsifiers that promote the formation of w/o emulsions. Polyethylene glycol sorbitan fatty acid esters (Tweens) are water-soluble emulsifiers that promote the formation of o/w emulsions. Pluronics are block copolymers of hydrophilic poly (oxyethylene) (POE) and hydrophobic poly(oxypropylene) (POP) represented by the general formula POEnPOPm-POEn, where n and m represent the number of OE and OP, respectively.
The Spans and the Tweens come in different molecular weight or size ranges, which differ in their physical properties.
| Trade name | Structure/name | Applications |
| Triton™ X-100 | Polyoxyethylene glycol octylphenol ethers: C8H17–(C6H4)–(O-C2H4)1–25–OH | Wetting agent – coatings |
| Nonoxynol-9 | Polyoxyethylene glycol alkylphenol ethers: C9H19–(C6H4)–(O-C2H4)1–25–OH | Spermacide |
| Polysorbate | Polyoxyethylene glycol sorbitan alkyl esters | Food ingredient |
| Span® | Sorbitan alkyl esters | Polishes, fragrance carriers |
| Poloxamers, Tergitol™, Antarox® | Block copolymers of polyethylene glycol and polypropylene glycol | Various |
Anionic surfactant
Anionic surfactants contain anionic functional groups at their head, such as sulfonate, phosphate, sulfate and carboxylates. Alkyl sulfates include ammonium lauryl sulfate, sodium lauryl and the related alkyl-ether sulfates sodium laureth sulfate, also known as sodium lauryl ether sulfate (SLES), and sodium myreth sulfate. These are the most common surfactants and comprise the alkyl carboxylates (soaps), such as sodium stearate. The stearates comprise >50% of the global usage of surfactants. Many of these find utilization in emulsion polymerization. Other anionic surfactants include dioctyl sodium sulfosuccinate (DOSS), perfluorooctanesulfonate (PFOS), linear alkylbenzene sulfonates (LABs) and perfluorobutanesulfonate, as well as alkyl-aryl ether phosphates. More specialized species include sodium lauroyl sarcosinate and carboxylate-based fluorosurfactants such as perfluoroconate (PFOA or PFO).
| Trade name | Structure/name | Applications |
| Pentex 99 | Dioctyl sodium sulfosuccinate (DOSS) | Wetting agent – coatings, toothpaste |
| PFOS | Perfluorooctanesulfonate (PFOS) | Scotchguard™, Skydrol™ |
| Calsoft® | Linear alkylbenzene sulfonates | Laundry detergents, dishwasher detergents |
| Texapon® | Sodium lauryl ether sulfate | Shampoos, bath products |
| Darvan® | Lignosulfonate | Concrete plasticizer, plasterboard, DMSO |
| N/A | Sodium stearate | Handsoap, HI&I products |
Cationic surfactant
Cationic surfactants are comprised of a positively charged head. Most of cationic surfactants find use as anti-microbials, anti-fungals, etc. in the cleaners industry (Benzalkonium chloride (BAC), Cetylpyridinium chloride (CPC), Benzethonium chloride (BZT)). The cationic nature of the surfactants is not typically consistent with the world of nonionic and anionic charges, and they disrupt cell membranes of bacteria and viruses. Permanently charged quaternary ammonium cations include: Alkyltrimethylammonium salts: cetyl trimethylammonium bromide (CTAB) and cetyl trimethylammonium chloride (CTAC).
Zwitterionic surfactants
Zwitterionic (amphoteric) surfactants have both cationic and anionic centers attached to the same molecule. The anionic part can be variable and include sulfonates, as in the sultaines CHAPS (3-[(3-Cholamidopropyl)dimethylammonio]-1-propanesulfonate). Betaines such as cocamidopropyl betaine have a carboxylate with the ammonium. The cationic part is based on primary, secondary, or tertiary amines or quaternary ammonium cations. Zwitterionic surfactants are often sensitive to pH and will behave as anionic or cationic based on pH. Fast dry (“coacervation”) latex traffic paints are based on this concept, with a drop in pH triggering the latex in the paint to coagulate.
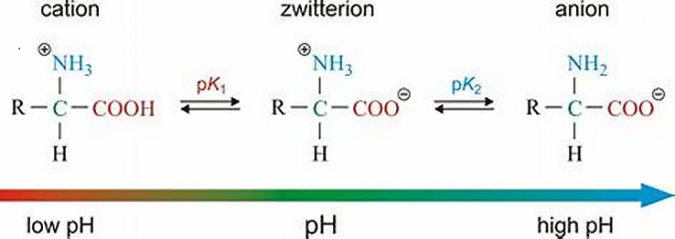
Amphoteric surfactants are zwitterionic molecules that are, at least at some pH value, both anionic and cationic at the same time so that the hydrophilic portion of the molecule has internally neutralized positive and negative charges.
Find surfactants, additives, binders and thousands of more materials at ULProspector.com!
Resources:
- https://aocs.onlinelibrary.wiley.com/hub/journal/15589293/forauthors.html
- Use of Dielectric Constants in the Classification of Surfactants https://jpharmsci.org/article/S0022-3549(15)33934-4/pdf
- Surfactant https://www.sciencedirect.com/topics/chemistry/surfactant
- Classification of surfactants due to structure of their molecules, https://www.researchgate.net/figure/Classification-of-surfactants-due-to-structure-of-their-molecules_fig1_319618703
- Lowering surface tension – Surfactants in coating materials https://www.ulprospector.com/knowledge/3106/pc-surface-active-agents-surfactants/
- https://www.ulprospector.com/knowledge/8516/pc-surfactant-infographic/
- https://www.ulprospector.com/knowledge/6954/pcc-how-to-select-natural-surfactants-video/
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



Excellent Marc.