 Biopolymers provide an important tool for sequestering carbon dioxide from the atmosphere, allowing mankind the ability to begin reversing global warming, as well as to improve our overall global environmental footprint. As polymer scientists learn more about how to leverage the performance properties that nature has already discovered, we expand our ability to make lighter and stronger vehicles, more bio-compatible medical devices, plastics that biodegrade when released back into nature and less toxic monomers to our environment. The following is a brief review of several of the most commercially significant biopolymers, with emphasis on thermoplastics.
Biopolymers provide an important tool for sequestering carbon dioxide from the atmosphere, allowing mankind the ability to begin reversing global warming, as well as to improve our overall global environmental footprint. As polymer scientists learn more about how to leverage the performance properties that nature has already discovered, we expand our ability to make lighter and stronger vehicles, more bio-compatible medical devices, plastics that biodegrade when released back into nature and less toxic monomers to our environment. The following is a brief review of several of the most commercially significant biopolymers, with emphasis on thermoplastics.
Polyamide 11
The chemical process of creating the Polyamide 11 biopolymer begins with castor oil, which typically contains between 84-94% ricinoleic acid. Castor oil is first transesterified with methanol to yield methyl ricinoleate. Methyl ricinoleate is then “cracked” to create heptaldehyde and methyl undecylenate. The methyl undecylenate is then hydrolyzed to release methanol and undecylenic acid. At this point, hydrogen bromide is added across the double bond of undecylenic acid to form the 11-bromo derivative followed by displacement with ammonia to form 11-amino undecanoic acid. In a final step, 11-amino undecanoic acid is then homo-polymerized to form Polyamide 11, leading to a 100% bio-based polymer.
The unique 11-carbon chain yields a crystallinity profile that is very high in hydrogen bond density (chemical structure below). This contributes to a high performance resin with higher melt point, lower fuel and gas permeability, improved impact properties and lower environmental footprint compared to the more traditional Polyamide 12. Polyamide 11 is typically more flexible, has better dimensional stability & better impact properties versus typical short chain polyamides such as Polyamide 6.
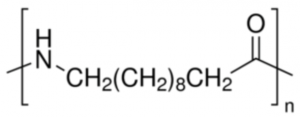
Polyamide11 is easy to process, using most processing technologies (extrusion, extrusion blow-molding, injection molding, 3D printing, powder coating and rotomolding). The polyamide11 matrix accommodates countless additives and filling agents, such as plasticizers, stabilizers, colorants, lubricants, impact modifiers, glass fiber, carbon fiber, etc. Polyamide 11 has a wide range of working temperatures (-40° C to +130° C). A table of general properties of Polyamide 11 versus Polyamide 6 are provided in the table below. High temperature, transparent or flexible derivations of Polyamide 11 are also available from suppliers, such as Arkema.
General properties of Polyamide 11 versus Polyamide 6
| Density (g/cm3) | Flexural modulus (MPa, dry) | Flexural modulus (MPa, conditioned) | Elongation (%) | Water absorption (%, 23°C/50%RH) | Melting point (°C) | Tg (°C) | |
| Nylon 11 | 1.03 | 1100-1300 | 1000-1200 | 300-400% | 0.8 | 189 | 42 |
| Nylon 6 | 1.14 | 2200 – 2400 | 1100 – 1300 | 300% | 3 | 210-220 | 48-60 |
As a thermoplastic polymer, Polyamide 11 is recyclable but is not biodegradable. Pragati is a sustainable castor initiative founded by Arkema, BASF, Jayant Agro-Organics LTD and Solidaridad, that partners with farmers in India where the majority of global castor beans are grown. The goal of the program is to help farmers improve yields and reduce environmental impacts, thus helping cultivate castor beans effectively and ecologically.
Polyamide 11 biopolymer is used in footwear, sporting equipment, ski components, textiles, brushes, automotive, medical, metal coatings, wire and cable jacketing, and electrical components. Due to its low water-absorption and subsequent dimensional stability as a result of this attribute and combined with heat, chemical and burst resistance and flexibility, Polyamide 11 is also used widely in tubing applications such as fuel lines, hydraulic hoses, air lines catheters and beverage tubing.
Bio-Polyethylene Terephthalate and Polyfuranoates
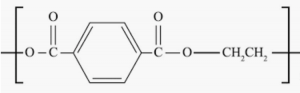
Polyethylene terephthalate biopolymer is an incredibly useful material, that has improved the state of humanity. It keeps our food fresh and allows for lightweight packaging that allows products to be shipped cheaply with reduced greenhouse gas emissions. It is used to package and construct medical devices, fabrics and threads. PET is under our bare feet in our carpet as we walk across the room. It makes up the fibers in our clothing, keeping us warm and wicks water away when we sweat. It is used to keep us comfortable in the form of fiber-filled pillows and furniture cushions. It is used to make films for electronic and oxygen barrier applications. There is no question that PET is a fantastic plastic that has made our lives better in many ways.
Coca Cola introduced PlantBottle™ technology in 2009, making it the world’s first fully recyclable PET plastic bottle produced using ethylene glycol derived from sugar cane. The ethylene glycol, supplied by India Glycol, and copolymerized with terephthalic acid, provided their PET bottles with 30% biorenewable content, and resulted in no change to the biopolymer’s chemical structure or properties. Coca Cola today claims that the CO2 reduction benefit provided by this bio-PET since its launch in 2009 is equivalent to removing nearly 1 million vehicles from the road.

In 2012, Coca-Cola, Ford Motor Company, H.J. Heinz Company, NIKE, Inc. and Procter & Gamble announced the formation of the Plant PET Technology Collaborative. This collaborative has sponsored a variety of research efforts aimed at making the terephthalic acid portion of the PET biorenewable, as well as the ethylene glycol.
The most notable efforts towards replacing petroleum-based PET with PET biopolymer include:
- Conversion of plant-derived butanol into para xylene via deoxygenation and dehydration at high temperatures and in the presence of catalyst using technology patented by Virent (US 8,962,902). This plant-based para-xylene could then be oxidatively converted to terephthalic acid for use in making bioPET.
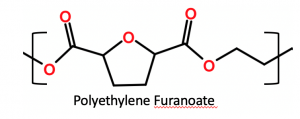
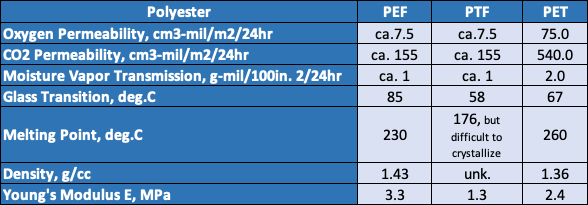
- Avantium’s production of polyethylene furanoate biopolymer (PEF, chemical structure below) from biobased ethylene glycol and sugar-based furan dicarboxylic acid. The resulting PEF is biobased, recyclable and has 10 times better barrier properties to oxygen than PET, 6-10 times better barrier properties to carbon dioxide and improved mechanical strength versus PET. Avantium has recently come to agreement with Mitsui Chemical and both companies intend to produce PEF for packaging applications.
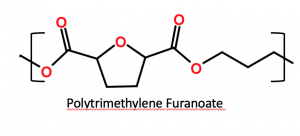
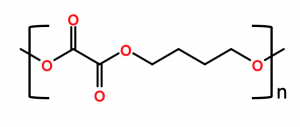
- A DuPont / ADM joint venture, in which polytrimethylene furanoate biopolymer (PTF, chemical structure below) is being produced in a pilot facility for bottle and other packaging applications. Again, PTF is biorenewable when produced using DuPont’s biorenewable 1,3-propane diol and sugar-based dimethyl furanoate. The resulting polymer has excellent barrier and physical properties and is recyclable.
A table comparing the properties of these biopolymers is provided below.
Polytrimethylene Terephthalate
Polytrimethylene terephthalate (PTT) biopolymer is an aromatic polyester thermoplastic that is prepared via the polymerization of 1,3-propanediol with terephthalic acid. The petroleum-based polymer was discovered in 1941 (US 2,465,319) but has only achieved commercial success recently through DuPont’s use of biorenewable 1,3-propanediol, which is obtained from the fermentation of sugars (US 6,013,494) using genetically modified organisms. The chemical structure of PTT is provided below.
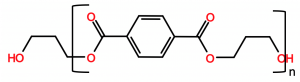
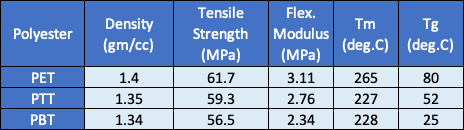
The biopolymer contains about 37% biobased content, due to the use of biorenewable 1,3-propanediol. A table comparing the physical properties of PTT to PET and polybutylene terephthalate is provided below.
DuPont’s Sorona PTT biopolymer has found utility in carpet fiber and textile applications, due to its durability and toughness, combined with its relatively soft feel.
Polybutylene Succinate
Polybutylene succinate (PBS) is produced by PTT MCC Biochem, which is a joint venture between PTT Public Company, Limited and Mitsubishi Chemical Corporation. PBS biopolymer is produced in Thailand using biorenewable succinic acid and biorenewable 1,4-butanediol, forming the chemical structure represented in the figure below.
One of the interesting features of PBS is its ability to biodegrade in the soil to form carbon dioxide and water under ambient conditions. This means that products made from PBS can be disposed of along with organic wastes to form a compostable mixture in an open-air landfill. PBS biopolymer has a density of 1.26 g/cc, a melting point of 115 deg.C, a tensile strength at break of 30 MPa, and an elongation of about 200%.
Bio-Polylactic Acid
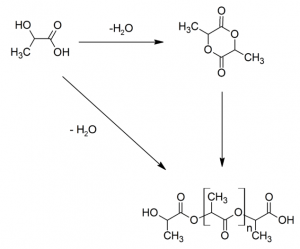
Polylactic acid (PLA, chemical structure below) biopolymer is based upon corn sugars and is produced via the ring-opening polymerization of lactide, or the polyesterification of lactic acid, as shown below.
The largest manufacturers of polylactic acid are NatureWorks (a joint venture between Cargill and PTT), WeforYou, Evonik and Total-Corbion (a joint venture between Total and Corbion.
Polylactic acid (PLA) has numerous applications including plastic films, bottles, and biodegradable medical devices that are expected to biodegrade within 6-12 months within the body. PLA biopolymer has recently also become extremely popular in 3D printing applications, due to the ease with which it melts and resolidifies during extrusion from the microextrusion head of a thermal printer.
PLA may be recycled and industrially composted, providing even more beneficial features relative to its environmental footprint.
Conclusions
We exist in an age where the world’s climate is at a tipping point – one that was created by man, and now must be solved, in part by polymer scientists who are reinventing how we produce valuable monomers for use in polymers critical to our survival and way of life. Some of our best minds are racing to develop biopolymers that sequester carbon dioxide from the atmosphere to begin the process of reversing climate change on our planet. This common goal is being embraced by scientists and engineers everywhere – not only those who are working on more sustainable bioplastics, but those who are creating our future modes of transportation, energy, building materials, agriculture and manufacturing.
Unlimited Premium Prospector members have access to the Biopolymer Advanced Search tool. Add time-saving tools to your Prospector account today!
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



Very nice overview! Complete newbie question: How does any of that “sequester CO2”? I was expecting to read of polymers that can scrub CO2 from the atmosphere, but the only mention of CO2 I saw was how some of these materials GENERATE it as they degrade. I must have missed it…
Hi Michael, Great question! Anytime you create a biorenewable monomer using a fermentation process, for example, the carbon (dioxide) used by the microbes or plants is sequestered into the resulting monomer. This sequestered carbon is transferred to the thermoplastic. Thanks and best regards, Rick Tabor