 The original article on surfactants was written in the fall of 2015 and updated in 2021. Although the general topic of surfactants has not changed in nearly 8 years, several new chemistries have surfaced, and there wasn’t nearly as much emphasis on bio-based or green chemistry back then, as there is today. Some of the chemistries are either considered in some way biologically incompatible, or in others, have a degree of toxicity. In addition, if you are new to coatings you should also be looking at ancillary markets such as personal care and cosmetics, since these markets use surfactants quite a bit, and you might find something in an adjacent market that works perfectly well in coatings and adhesives. Personal care surfactants often have the same chemistry as paint surfactants, but perhaps different names or slightly different functions. In comparing surfactants, you can always look at the CAS to ascertain equality.
The original article on surfactants was written in the fall of 2015 and updated in 2021. Although the general topic of surfactants has not changed in nearly 8 years, several new chemistries have surfaced, and there wasn’t nearly as much emphasis on bio-based or green chemistry back then, as there is today. Some of the chemistries are either considered in some way biologically incompatible, or in others, have a degree of toxicity. In addition, if you are new to coatings you should also be looking at ancillary markets such as personal care and cosmetics, since these markets use surfactants quite a bit, and you might find something in an adjacent market that works perfectly well in coatings and adhesives. Personal care surfactants often have the same chemistry as paint surfactants, but perhaps different names or slightly different functions. In comparing surfactants, you can always look at the CAS to ascertain equality.
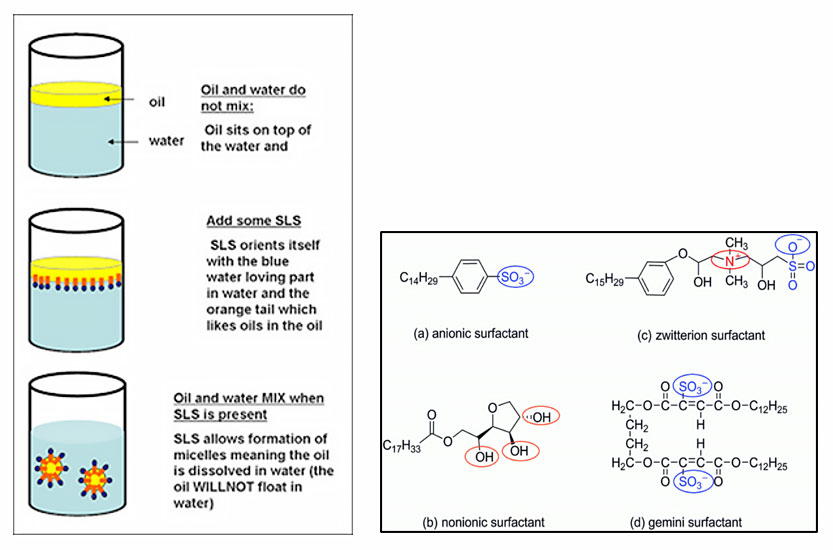
Prospector® Knowledge Center has an excellent expert in Belinda Carli for Personal Care & Cosmetics. See her article entitled “How to Select Natural Surfactants (video)”, or one from 2021, “Sustainability and Carbon Neutral Beauty”. A natural surfactant has to have both the head and tail groups to come from truly natural sources. Another article from another author that pulls it all together is “Breaking the Tension with Surfactants”.
In addition to industrial applications, surfactants have the potential to overcome the current limitations in nanotechnology. For example, amphiphilic surfactants have been reported as stabilizers for the preparation of stable dispersions of hydrophobic inorganic nanomaterials such as carbon nanotubes, graphene and transition metal dichalcogenides. Since the first graphene exfoliation method, micromechanical exfoliation, was discovered in 2004, researchers have attempted scale-up of the process by stabilizing exfoliated graphene in solvents. Considering the accessibility and environmental toxicity of organic solvents, water is an ideal solvent system for preparing nanomaterial dispersions, but most inorganic nanomaterials have hydrophobic surfaces and thus exhibit repellent behavior in water.
As far as bio-based surfactants, in 2020, thirty percent of global respondents were willing to pay a premium for products that deliver on social accountability claims (Nielsen, 2020). Bio-based surfactants are designated by the new EN17035. The chemical industry is now able to use a transparent definition and division which also enables it to communicate in a comparable way to the end-user. Companies such as BASF, Ethox, Solvay, Dow Chemical and Locus Fermentation Solutions, among many others, are producing more biosurfactants. Almost all surfactant companies that manufacture anionic and non-ionic products offer biodegradable or other green surfactants, that in the US meet the US EPA’s safer choice standard for residential, institutional, industrial, and janitorial cleaning products.
Bio-based surfactants are derived primarily from natural sources (i.e. both the head and tail molecular group). The more common types of bio-based surfactants are those with carbohydrate head groups, where alkyl polyglucosides (APGs) and sucrose esters (SEs) lead this sub-category. As global regulations and user mandate for sustainability and safety increase, evidence to further support these bio-based surfactants as alternatives to their petrochemical counterparts is advantageous. Use of the green chemistry structure is a suitable way to do this. While many of the discussed principles are enforced industrially, others have only yet been applied at a laboratory scale or are not apparent in literature.
Surfactants play an important role as dispersing, emulsifying, cleaning, wetting, foaming and anti-foaming agents in many practical applications and products, including paints, emulsions, adhesives, firefighting (foams), inks, biocides (sanitizers), shampoos, toothpaste, detergents, insecticides, deinking of recycled papers, ski waxes, and many others. Since this was written, firefighting foams that utilize PFAS (poly-fluoroalkyl substances) have come under extreme scrutiny since fluorinated compounds are considered forever chemicals and don’t break down in the environment.
The dynamic forces of surfactant adsorption is of great importance for practical applications such as in emulsifying or coating processes as well as foaming, where bubbles or drops are swiftly generated and need to be stabilized. As the interface is created, the adsorption is hampered by the diffusion of the surfactant to the interface, which can result in the kinetics being limited. These energy barriers can be due to electrostatic or steric repulsions; steric repulsions form the basis of how dispersants work. Surface rheology of surfactant layers is important to the stability of foams and emulsions.
A primer of the surfactant types and chemistries follows but look at past UL articles for more in-depth information. Surfactants are materials that lower the surface tension (or interfacial tension) between two liquids or between a liquid and a solid. In the general sense, any material that affects the interfacial surface tension can be considered a surfactant, but in the practical sense, surfactants may act as wetting agents, emulsifiers, foaming agents, and dispersants, among others.
There are 4 types of surfactants with a brief review of each as follows. These classifications are based on the composition of the polarity of the head group: nonionic, anionic, cationic, amphoteric. There are some companies that are claiming additional types based on different chemistries. One such company is LanKem and the type of surfactant is a BioLoop surfactant.
Nonionic surfactant
A non-ionic surfactant has no charge groups in its head. The head of an ionic surfactant carries a net charge. If the charge is negative, the surfactant is more specifically called anionic; if the charge is positive, it is called cationic. If a surfactant contains a head with two oppositely charged groups, it is termed zwitterionic. Commonly encountered surfactants of each type are listed as follows. A complete compendium can be found on www.ULProspector.com.
Anionic surfactant
Anionic surfactants contain anionic functional groups at their head, such phosphate, sulfate as sulfonate, and carboxylates. Alkyl sulfates include ammonium lauryl sulfate, sodium lauryl and the related alkyl-ether sulfates sodium laureth sulfate, also known as sodium lauryl ether sulfate (SLES), and sodium myreth sulfate. These are the most common surfactants and comprise the alkyl carboxylates (soaps), such as sodium stearate. The stearates comprise >50% of the global usage of surfactants. Many of these find utilization in emulsion polymerization. Other anionic surfactants include dioctyl sodium sulfosuccinate (DOSS), linear alkylbenzene sulfonates (LABs), as well as alkyl-aryl ether phosphates.
Cationic surfactant
Cationic surfactants are comprised of a positively charged head. Most cationic surfactants find use as anti-microbials, anti-fungals, etc. in household, institutional and industrial cleaners (Benzalkonium chloride (BAC), Cetylpyridinium chloride (CPC), Benzethonium chloride (BZT)). The cationic nature of the surfactants is not typically consistent with the world of non-ionic and anionic charges, and they disrupt cell membranes of bacteria and viruses. Permanently charged quaternary ammonium cations include Alkyltrimethylammonium salts: cetyl trimethylammonium bromide (CTAB) and cetyl trimethylammonium chloride (CTAC).
Zwitterionic surfactants
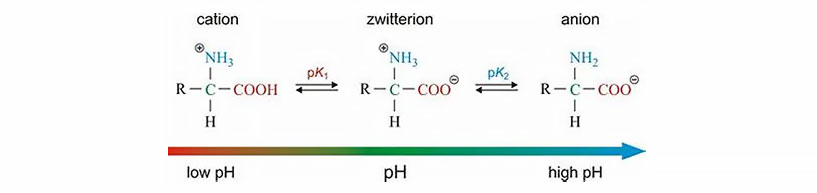
Zwitterionic (amphoteric) surfactants have both cationic and anionic centers attached to the same molecule. The anionic part can be variable and include sulfonates, as in the sultaines CHAPS (3-[(3-Cholamidopropyl)dimethylammonio]-1-propanesulfonate). Betaines such as cocamidopropyl betaine have a carboxylate with the ammonium. The cationic part is based on primary, secondary, or tertiary amines or quaternary ammonium cations. Zwitterionic surfactants are often sensitive to pH and will behave as anionic or cationic based on pH. Fast dry (“coacervation”) latex traffic paints are based on this concept, with a drop in pH triggering the latex in the paint to coagulate.
[i] How to select natural surfactants for personal care products (ulprospector.com)
[ii] Sustainability and Carbon Neutral Beauty – Prospector Knowledge Center (ulprospector.com)
[iii] Surfactants in paints: how they work and current market trends | Prospector (ulprospector.com)
[iv] Lankem Surfactants | Biobased | United Kingdom
[v] 119-01491-01-dow-surfactants-selection-guide.pdf
[vi] A review on the synthesis of bio-based surfactants using green chemistry principles | SpringerLink
Resources
- https://aocs.onlinelibrary.wiley.com/hub/journal/15589293/forauthors.html
- Surfactant
- Classification of surfactants due to structure of their molecules
- How to Select Natural Surfactants [VIDEO]
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.


