 Polyisocyanates and polyurethanes have been the subject of several previous articles in Prospector, the most recent articles were available in 2019 and 2021. This article will concentrate primarily on polyisocyanates and PU technology rather than the reaction products of isocyanates with functionalities other than hydroxyl. Over the last decade, there has been a plethora of more sustainable, ecofriendly and less toxic polyisocyanate and polyurethane chemistries developed. These include those derived from biobased sources (rather than from petrochemicals) or the use of nonisocyanate (NIPU) technologies, or polyisocyanates containing very low levels of free isocyanate.
Polyisocyanates and polyurethanes have been the subject of several previous articles in Prospector, the most recent articles were available in 2019 and 2021. This article will concentrate primarily on polyisocyanates and PU technology rather than the reaction products of isocyanates with functionalities other than hydroxyl. Over the last decade, there has been a plethora of more sustainable, ecofriendly and less toxic polyisocyanate and polyurethane chemistries developed. These include those derived from biobased sources (rather than from petrochemicals) or the use of nonisocyanate (NIPU) technologies, or polyisocyanates containing very low levels of free isocyanate.
Polyisocyanates provide enhanced performance and multiple options for crosslinking with a variety of reactive groups which contain an active hydrogen:
- Hydroxy
- Amino
- Imino
- Ketimine
- Carboxyl (forms CO2)
- Urethanes
- Ureas
- Acetoacetylated resins
This article will feature primarily polyisocyanates and PU technology rather than the reaction products of isocyanates with functionalities other than hydroxyl. Reactive polyisocyanates are defined as any compound containing multiple isocyanate functional groups as well as any polymer prepared from an isocyanate, especially a polyurethane. Polyisocyanates are used in a variety of coating and adhesives applications as building blocks for higher molecular weight polyisocyanates, blocked isocyanates and polyurethane polymers such as solvent-born or waterborne polyurethane dispersions PUD).
NIPU
Two-component isocyanate-containing chemistries as a class are considered to be less toxic and more environmentally acceptable. Accordingly, NIPU technology as a way to provide a polyurethane coating has generated much attention in the last decade.
Two-component NIPU ambient cure automotive refinish primer surfacer coating technology is based on the reaction between polycarbamates and polyaldehydes. Dow Chemical reports the ability to decouple cure speed from pot life with this technology to provide fast cure to enable sanding, excellent solvent resistance, adhesion and humidity resistance1. A later article2 presented at the American Coatings Show indicates that this chemistry also provides improved appearance along with stain and chemical resistance and exterior durability when applied over wood.
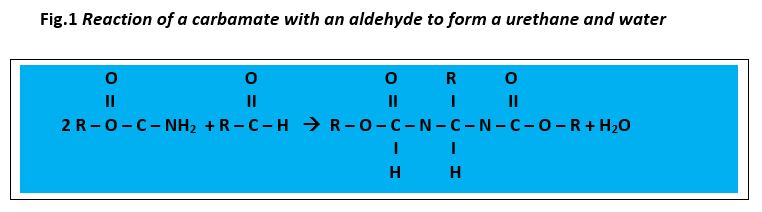
Two-component NIPU high solids ambient cure coating prototypes were also developed utilizing cyclic carbonate/amines to form polyurethane polyamines cured with aliphatic epoxy functional hardeners for advanced rain-erosion resistance. Although the technology does not meet the criteria for advanced rain erosion for military applications, with additional optimization, the core technology platform may also have utility for other applications such as those for general industrial, transportation and product finishing.
2HN –[R1] – NH2 + polycyclic carbonate à 2HN –[R2] – NH2 + aliphatic epoxy -> crosslinked film
R1 = aliphatic or acyclic R2 = polyurethane polyamine (NIPU)
Biobased and Sustainable Polyisocyanates
Eco N 7300 is an isocyanate functional trimer from Covestro that contains 70% renewable carbon content derived from starch in field corn. The trimer is based on pentamethylene diisocyanate (PDI).
BASF also has a low carbon footprint MDI (methylene diphenyl diisocyanate) Lupranat ZERO (Zero emission, Renewable origin) made from biobased raw materials that have a Product Carbon Footprint (PCF) of zero.
Poly Isocyanates are divided into two major classes, aromatic and aliphatic, although aromatic isocyanates react faster than do aliphatic-based isocyanates they are used in fewer applications as they are unstable when exposed to sunlight and air resulting in oxidative degradation and yellowing. The predominate isocyanates used as building blocks to form isocyanate prepolymers are aliphatic in nature (see fig. 2) and include HDI, IPDI and H12MDI.
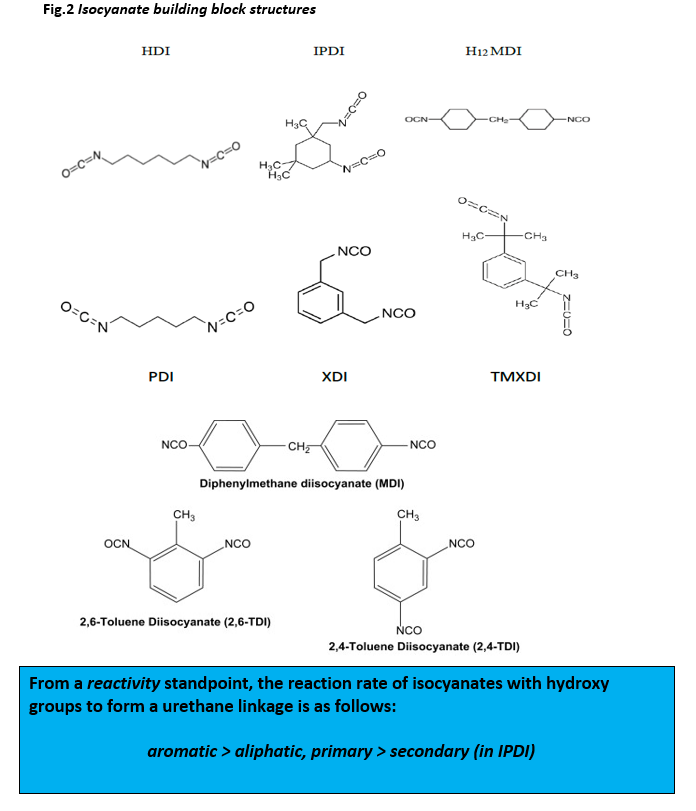
For example, HDI can be used to form NCO functional prepolymers with 2 NCO groups – uretdione (4 member heterocycle), allophanate or carbodiimide or with 3 NCO groups – isocyanurate (6 member heterocycle), biuret, or an imminoxadiazinedione. The trimerization of HDI can be catalyzed by a number of Lewis bases of inorganic and organic salts as well as other materials. The ratio of trimer to oligomer is used to adjust the molecular weight and viscosity. The advantage of a cyclic trimer is that a six-membered ring is chemically and thermally very stable. Accordingly, trimers are made from not only HDI, but IPDI and TDI as well. TMXDI-based polyisocyanates are used in waterborne coatings and adhesives, whereas IPDI-based uretdiones are often used in TS powder coatings. The four-member uretdione ring is not as stable as a six-membered ring and thus various substitutions on the uretdione can be used to alter the reactivity and stability.
Isocyanate-terminated prepolymers
Isocyanate-terminated prepolymers or hydroxy-terminated polyurethanes can be synthesized by using either an excess stoichiometric amount of diol with diisocyanate to form a hydroxy-terminated polyol or conversely using an excess amount of diisocyanate to form an isocyanate-terminated prepolymer and then removing the excess diol or isocyanate respectively.
Single-component thermosetting polyurethanes can be formulated by using blocked isocyanates. Blocked isocyanates are heat activated at different temperatures to regenerate the reactive isocyanate for crosslinking with the functional polymer (normally hydroxy-functional) to form a urethane linkage. The blocking agent is volatilized at the activation temperature.
For unblocking temperatures, from the lowest to highest (left to right):
Dimethylmaleate < Dimethylpyrazole < Methyl ethyl ketoxime < e-Caprolactone
Polyisocyanate suppliers listed in Prospector:
- BASF
- Covestro
- Evonik
- Keeneyes International Co, Ltv
- Kowa American Corp.
- S.A.P.I.C. SpA
- T&L Co., Ltd
- Tosoh Corp
- Vencorex
- Wanhua Chemical Group Co., Ltd
- Yantai Suny Chem International Co., Ltd
References
- Coatings Tech/February 2017/Vol.14, No 2
- ACA presentation, Dow Chemical 2023
- Progress in Organic Coatings, Volume 165, April 2022, 106728
- https://apps.dtic.mil/sti/trecms/pdf/AD1191459.pdf
- https://chemicals.basf.com/global/monomers/documents-/20792049_OnePager_LupranatZERO_EN_final.pdf
- Polyurethanes for Coatings & Adhesives – Chemistry & Applications, F.E Gollig et.al. Covestro AG
- Organic Coatings, Science and Technology, Volumes 1,2,3 and 4
- Polyisocyanates Deep Dive – Update Marc Hirsh
- Get a Reaction with Urethane Coatings Ron Lewarchik
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.


