 The global acrylic coating market size is estimated to be approximately 60,000 million in USD with CAGR of over 4% until 2030. Coatings utilizing acrylic resins and oligomers are the leading polymer technology in the coatings industry, being utilized in large market segments including architectural, automotive OEM, automotive refinish, traffic marking coatings, floor finishes and product finishes. Major applications include use in waterborne, solvent borne, powder and light cure coatings and contain one or more acrylate, methacrylate and/or other vinyl monomers to synthesize a copolymer or terpolymer. Acrylic polymers as a class provide good weather resistance, resistance to hydrolysis, gloss and color retention in exterior applications. Due to their versatility and performance, acrylic coatings account for over 25% of all coatings with global sales estimated to approach $25 billion. Acrylic resins and acrylate functional materials can be thermoplastic or thermoset and are used in organic solvent born, waterborne, powder and radiation-curable coatings.
The global acrylic coating market size is estimated to be approximately 60,000 million in USD with CAGR of over 4% until 2030. Coatings utilizing acrylic resins and oligomers are the leading polymer technology in the coatings industry, being utilized in large market segments including architectural, automotive OEM, automotive refinish, traffic marking coatings, floor finishes and product finishes. Major applications include use in waterborne, solvent borne, powder and light cure coatings and contain one or more acrylate, methacrylate and/or other vinyl monomers to synthesize a copolymer or terpolymer. Acrylic polymers as a class provide good weather resistance, resistance to hydrolysis, gloss and color retention in exterior applications. Due to their versatility and performance, acrylic coatings account for over 25% of all coatings with global sales estimated to approach $25 billion. Acrylic resins and acrylate functional materials can be thermoplastic or thermoset and are used in organic solvent born, waterborne, powder and radiation-curable coatings.

The Fox equation provides an estimate of the Tg of a polymer and can be calculated as the sum of the weight fractions of the individual monomers in the copolymer. Tg (1), Tg(2), Tg(3)….etc. are the Tg in degrees Kelvin of their high molecular weight homopolymers.

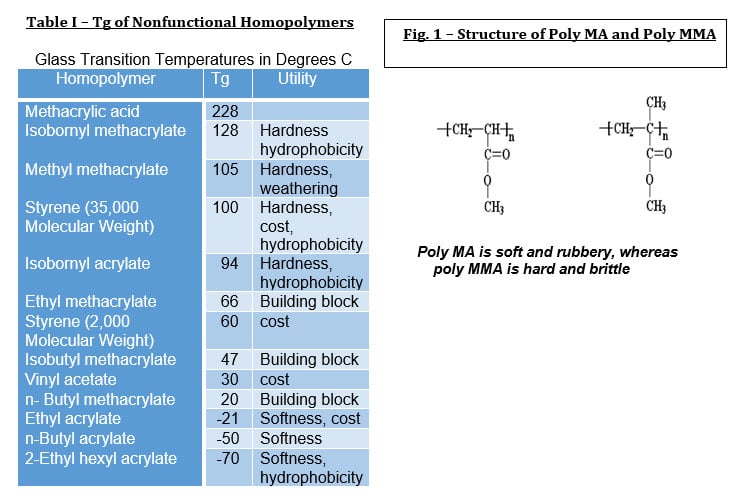
As Table 1 suggests, the glass transition temperature of the monomers selected for synthesis of a resin can be selected to enhance multiple properties that may include weather resistance, moisture resistance, resistance to oxygen permeation, flexibility, reactivity, cure and hardness. Most acrylic copolymers are comprised of three or more different monomers that are selected for the desired performance. For example, methyl methacrylate is commonly used to adjust the desired glass transition temperature (Tg), whereas BA and 2-EHA are used to provide hydrophobicity and to lower the Tg. Isobornyl Methacrylate is an anomaly as it provides a low viscosity even though the Tg of its homopolymer is high and is used as a partial replacement for MMA. IBMA is used in resins for automotive clearcoats. Methacrylic acid and acrylic acid provide polarity and hydrophilicity and can also improve adhesion to metal. Methacrylic and acrylic acid are used in the 1 – 2% range in emulsion formulations, at much higher levels these monomers increase the water solubility of the acrylic polymer.
Three broad classes of liquid coatings utilizing acrylic resins include thermoplastic, thermoset and waterborne. Acrylic resins can be synthesized by numerous methods; including RAFT (Radiation-induced controlled polymerization, FRP (Free Radical Polymerization) or ATRP (Atom Transfer Radical Polymerization), and for Waterborne acrylic resins, the polymerization methods include emulsion, solution, bulk and suspension. Whereas powder coatings utilize solid acrylic resins in powder form. These include acrylics functionalized with Glycidyl Methacrylate to crosslink with a dicarboxylic acid such as dodecanedioic acid or a carboxylic acid functional resin. Carboxyl functional acrylic powder coatings can also be cured with hydroxyalkyl amides. Lastly, some powder coatings also employ hydroxy functional acrylics for crosslinking with a blocked isocyanate or glycoluril.
Multiple polymeric architectures can be achieved with acrylic polymers and including graft, block, random, star, brush, hyperbranched and functional polymers.
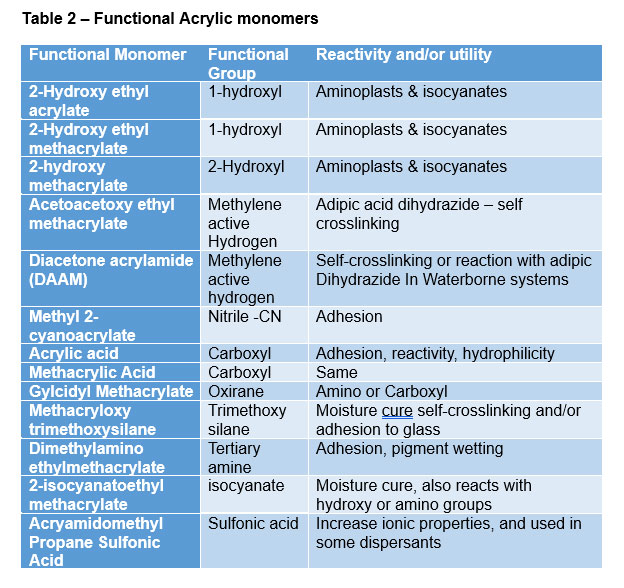
Acrylics can also be functionalized (Table 2) with a variety of monomers to provide additional performance benefits. As Table 2 indicates, being able to functionalize an acrylic resin with a wide range of functional moieties offers the ability to tailor the performance of the copolymer to provide improved adhesion over a variety of substrates, improved pigment wetting; and/or the ability to crosslink with a suitable reactant like an amino or isocyanate functional crosslinker in a single or two-component system. Other acrylic monomers are also available to impart sulfonic acid, or phosphoric acid functionality.
Functional monomers are used for multiple purposes which may include one or more of the following to enhance performance in ambient cure or thermoset coatings:
- Crosslinking in single or two package coatings
- Self-crosslinking
- Moisture cure
- Adhesion
- Pigment dispersion
- Increased or decreased polarity
- Moisture resistance
Thermosetting acrylic resins (TSA) are modified with functional monomers for crosslinking. They can be liquid or powder form and are designed with functional monomers to either react with themselves when exposed to heat or moisture, or with a cross-linker like melamine or an isocyanate to form a cross-linked film. Thermoset resins as a group can be lower molecular weight and thus have higher application solids than thermoplastic acrylics. Once cross-linked, as a class they offer films with excellent resistance to organic solvents, moisture and UV light and do not soften appreciably when exposed to moderately high temperatures as thermoplastics do. Carbamate functional acrylics can also be made for example by reacting an isocyanate functional acrylic with hydroxypropyl carbamate. Many of the acrylics in the category of functionalized acrylic resins are used in automotive OEM and refinish clearcoats to provide an excellent combination of mar resistance, chemical resistance and light stability.
Acrylic polymers are also used extensively to make water reducible and emulsion resins. Water reducible acrylic resins typically have acid numbers of 40 to 60. An example of a water reducible TSA would have a resin composition comprised of MMA/STY/BA/HEMA/AA in a weight ratio of 40/20/22/10/ 8 prepared by free radical polymerization in a water reducible solvent such as a glycol ether solvent like 2-butoxy ethanol with butyl alcohol. To begin the process of making paint, a suitable amine is used to neutralize the acrylic acid in the resin backbone to provide an acid salt. The amine is used at less than the theoretical level of neutralization using for example 2-(dimethyl amino) ethanol (DMEA) or 2-Amino-2-methyl-1-propanol (AMP). The next step in the paint-making process involves the addition of water.
Acrylic emulsion polymerization is carried out in water with monomers, a water-soluble initiator and surfactants. Many of the same monomers are used in emulsion polymerization as that in solution polymerization, however, the properties of the latex can be greatly affected by changes in the: polymerization conditions, monomer structure and solubility, monomer concentration, surfactant type and level, temperature, initiator type and initiator concentration are all variables that can affect the emulsion particle size, structure and molecular weight of the final polymer. A detailed examination of latex polymerization is a significantly large subject and is thus out of scope for this article. Acrylic polymers are also used in powder coatings along with acrylate functional acrylic resins for radiation cure coatings. The fundamental influence of monomer Tg, molecular weight and functional monomers remain the same for all the applications described herein.
In summary, it is the goal of this article to provide a heightened understanding of acrylic resin technology and its ramifications on paint performance characteristics and mechanical properties.
For additional information, please refer to the previous Prospector articles concerning waterborne resins, fundamentals of acrylic resins and waterborne coatings.:
Resources
Fundamentals of Waterborne Resin Technology
Flow, Leveling & Viscosity Control in Waterborne Coatings
Acrylic Resin Fundamentals
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.


