The optimization of shear-thinning properties of paints is more important for application properties than is shear thickening. The paint follows the viscosity profile as shear is increased, according to the predefined shear rate profile of the application. It is useful to first comprehend why shear thinning occurs so that one may recognize the cause and factors that influence the opposite shear profile that is a non-linear increase in viscosity as shear rate increases. This relationship between shear rate and shear stress helps to gain the necessary knowledge to modify formulations to improve application properties.
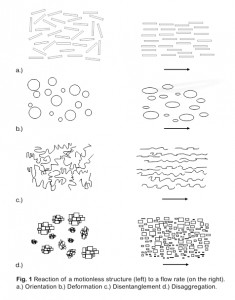
Dispersions respond by four mechanisms to shear rates (Fig. 1[1]) that reduce the resistance to flow. This figure illustrates the structural ordering observed upon induction of flow, such as impact of a shear rate.
Dilatant systems react – especially at high shear rates- by an increase of volume (formation of holes) with a tremendous increase in viscosity by a “sucking in“ of the fluid[2]. Simultaneously, particles move very closely to each other. Dilatants are non-Newtonian.
Another mechanism is somewhat similar to the behaviour of tensides, which build overstructures at high concentrations beyond their critical micelle concentration (CMC). Hard particles form structures as well[3].
Dilatancy Measurement
In order to calculate the dispersion composition in an agitator bead mill, to optimize both economically and efficiently and prevent equipment damage, the virtual ideal flow point is determined using the Daniel Titration (Fig. 2). This investigation of several binder/solvent mixtures (e.g., water as solvent) permits determination of wet- and flow- points (WP and FP, respectively ) as well as the DDI: Daniel Dilatancy Index. The following classification is in use in practice: 5-15% DDI, – tacky, poorly mixable, strongly dilatant; 15-30% DDI, somewhat dilatant, somewhat tacky; >30% DDI, not dilatant, poor mixability, very tacky. The wet point (WP) corresponds to the overall consistency of the mass, and is similar to the oil number test. The flow point (FP) indicates the extra volume of binder mixture per mass of solid necessary to permit the mass to just drop from a rod.

Dilatant Examples
- Highly concentrated (colloidal and non- colloidal) suspensions
- Hard particles (i. e. PVC Plastisole, ceramic inks)
- Hard particles in hard dispersions (i. e. quartz in acrylate (EU) dispersions of a high TG)
- Particles with high aspect ratio in high concentrations (plate-like effect pigments, lamellar functional pigments)
Modifications focus on achieving a deformable zone for hard particles: (1) By a different particle design with sterically effective surface treatments (in effect a steric increase in volume); or (2) By selecting combinations of sterically and electrostatically stabilizing dispersion or wetting agents respectively (also in solvent-borne paints). An example of rheology modifiers includes tailored wax dispersions that literally embed the flakes and ease processability (easy stir-up of sediments and ideal orientation) through controlling the rheological behaviour (lubricant-like behaviour). A second approach is core/shell technology for dispersions where the soft shell is a part of the particle polymer.
Measures for Dilatant Formulations

Functional and effect pigments orient parallel to the interfaces and form a ‘carpet’ parallel to the surface of the cured paint. Lamellar functional pigments such as Minatec® Grades (Merck), used in light conductive primers for electrostatic finishing of plastic car parts, need to build a percolation connection by particle contact.
Such pigments are wetted in low concentration and added for completion as a slurry. Their property ‘to stand in their own way’ generates sedimentation that is promoted by a high density. Dilatancy shows only at concentrations generally greater than 40 wt.%, but can be observed upon manual stirring (Fig. 3). Dilatancy is an important requirement for the formulation of free flowing pigment pastes. For examples, see raw material solutions below.
A partial list of examples of raw materials:
Binders: Core/Shell Technology for Inkjet applications RayRez® 165, RayCryl® 708E: Specialty Polymers
Wetting and Dispersion Aids: (Disperbyk® 182 (EU): Byk Altana (EU), EFKA® PX 4732 (EU): BASF SE (EU), Additol® XL 6509/100: Allnex (EU)
Special additives for lamelar / plate-like pigments
S/B: Ceratix® 8466 (EU), Cerafak® 100 (EU), W/B: Aquatix® 8421 (EU): Byk Altana
[1] (following) Introduction to Rheology and Rheometry, Gebhard Schramm, 2004, Thermo Electron
[2] “Flow properties of hard structured particle suspensions”, Smith, Zukoski, Journal of Rheology, 2004, 48, p. 1375
[3] CMC Critical Micelle Concentration *not needed here*
Search coatings materials on Prospector now…
About the Author:
 Born in 1961, Christina Kremers holds academic degrees in Organic Chemistry from the Universities of Duisburg and McMaster University, Canada. The doctorate was 1994 about a fundamental research of the photocatalytic degradation mechanism of special organic drinking water pollutants with anatase titanium dioxide. Before self-employment in 2004, she was besides further responsibilities, several years as head of quality assurance in charge for cycle time and failure reduction of paints at a known European Paints Manufacturer and before at an epoxy resin manufacturer in the same position. Coatings Science Lab is a technology consultancy with its own laboratory.
Born in 1961, Christina Kremers holds academic degrees in Organic Chemistry from the Universities of Duisburg and McMaster University, Canada. The doctorate was 1994 about a fundamental research of the photocatalytic degradation mechanism of special organic drinking water pollutants with anatase titanium dioxide. Before self-employment in 2004, she was besides further responsibilities, several years as head of quality assurance in charge for cycle time and failure reduction of paints at a known European Paints Manufacturer and before at an epoxy resin manufacturer in the same position. Coatings Science Lab is a technology consultancy with its own laboratory.
Coatings Science Lab eK, Wuppertal, Germany
www.coatings-science-lab.de
contact@coatings-science-lab.de
T +49 202 94600 850
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.



Leave a Reply or Comment